How To Calculate Root Mean Square Velocity
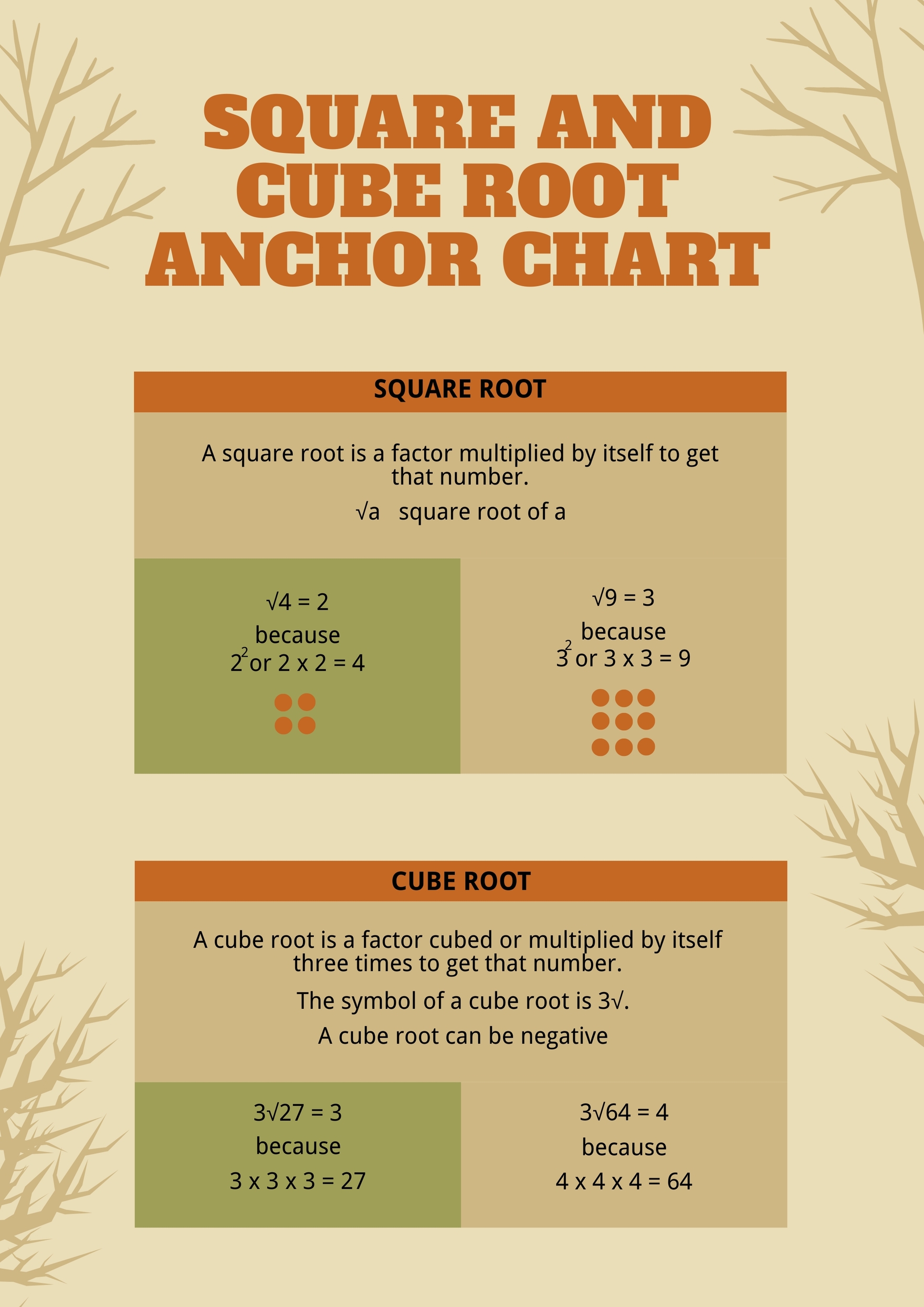
How To Calculate Root Mean Square Velocity - How do i calculate root mean square velocity? Why is root mean square velocity important in thermodynamics and. Vᵣₘₛ = √(3 * r * t / m), where r is the gas constant, t is the temperature, and m is the molar mass. Our root mean square speed calculator gives you an effortless way to calculate the rms speed for an ideal and mostly monoatomic gases. Use the root mean square velocity calculator to find the velocity of a particle in a gas based on the kinetic theory of gases. To calculate, we need to: Μ rms = (3rt/m) ½ Woosh, your result is instantly computed! This online chemistry calculator may be used to calculate root mean square velocity, mean velocity (a.k.a average velocity) and median velocity. The root mean square velocity (rms velocity) is a way to find a single velocity value for the particles.
Use the root mean square velocity calculator to find the velocity of a particle in a gas based on the kinetic theory of gases. Woosh, your result is instantly computed! The average velocity of gas particles is found using the root mean square velocity formula: To calculate, we need to: Μ rms = (3rt/m) ½ This online chemistry calculator may be used to calculate root mean square velocity, mean velocity (a.k.a average velocity) and median velocity. The root mean square velocity (rms velocity) is a way to find a single velocity value for the particles. Why is root mean square velocity important in thermodynamics and. Vᵣₘₛ = √(3 * r * t / m), where r is the gas constant, t is the temperature, and m is the molar mass. How do i calculate root mean square velocity?
To calculate, we need to: This online chemistry calculator may be used to calculate root mean square velocity, mean velocity (a.k.a average velocity) and median velocity. Our root mean square speed calculator gives you an effortless way to calculate the rms speed for an ideal and mostly monoatomic gases. Use the root mean square velocity calculator to find the velocity of a particle in a gas based on the kinetic theory of gases. Vᵣₘₛ = √(3 * r * t / m), where r is the gas constant, t is the temperature, and m is the molar mass. The root mean square velocity (rms velocity) is a way to find a single velocity value for the particles. Why is root mean square velocity important in thermodynamics and. The average velocity of gas particles is found using the root mean square velocity formula: How do i calculate root mean square velocity? Μ rms = (3rt/m) ½
34+ Root Mean Square Velocity Calculator OlufemiSalymat
Why is root mean square velocity important in thermodynamics and. The average velocity of gas particles is found using the root mean square velocity formula: Woosh, your result is instantly computed! To calculate, we need to: Μ rms = (3rt/m) ½
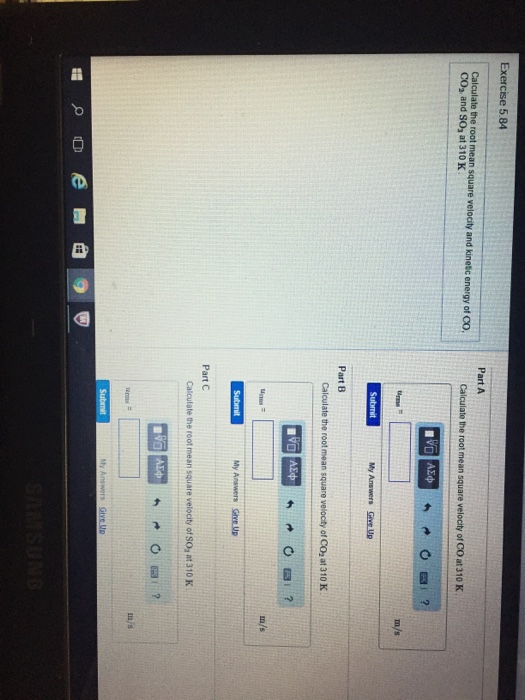
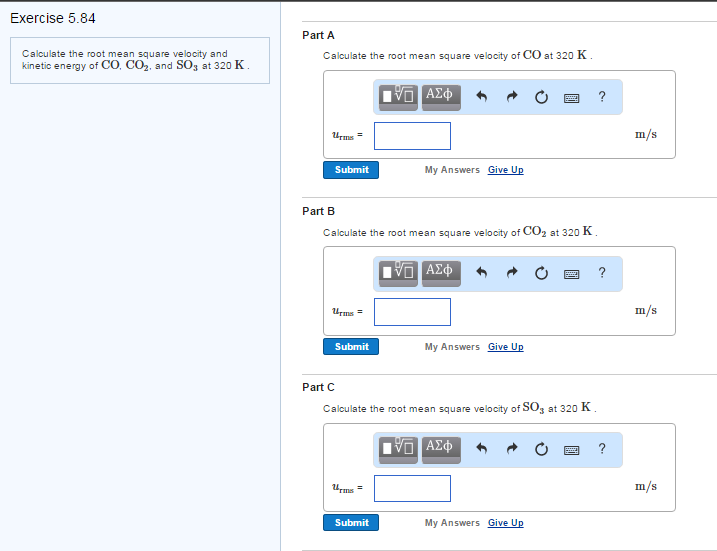
Calculate (a) The mean velocity, (b) The root mean square velocity and th..
Why is root mean square velocity important in thermodynamics and. The average velocity of gas particles is found using the root mean square velocity formula: Vᵣₘₛ = √(3 * r * t / m), where r is the gas constant, t is the temperature, and m is the molar mass. This online chemistry calculator may be used to calculate root.
Root Mean Square Velocity Calculator
Woosh, your result is instantly computed! Why is root mean square velocity important in thermodynamics and. How do i calculate root mean square velocity? The average velocity of gas particles is found using the root mean square velocity formula: Vᵣₘₛ = √(3 * r * t / m), where r is the gas constant, t is the temperature, and m.
Fine Beautiful Root Mean Square Velocity Derivation Pdf What Is
Our root mean square speed calculator gives you an effortless way to calculate the rms speed for an ideal and mostly monoatomic gases. Woosh, your result is instantly computed! Μ rms = (3rt/m) ½ How do i calculate root mean square velocity? This online chemistry calculator may be used to calculate root mean square velocity, mean velocity (a.k.a average velocity).
Fine Beautiful Root Mean Square Velocity Derivation Pdf What Is
How do i calculate root mean square velocity? Woosh, your result is instantly computed! Why is root mean square velocity important in thermodynamics and. Use the root mean square velocity calculator to find the velocity of a particle in a gas based on the kinetic theory of gases. The root mean square velocity (rms velocity) is a way to find.
Fine Beautiful Root Mean Square Velocity Derivation Pdf What Is
This online chemistry calculator may be used to calculate root mean square velocity, mean velocity (a.k.a average velocity) and median velocity. Use the root mean square velocity calculator to find the velocity of a particle in a gas based on the kinetic theory of gases. The root mean square velocity (rms velocity) is a way to find a single velocity.
Solved Calculate the root mean square velocity and
The average velocity of gas particles is found using the root mean square velocity formula: This online chemistry calculator may be used to calculate root mean square velocity, mean velocity (a.k.a average velocity) and median velocity. How do i calculate root mean square velocity? Use the root mean square velocity calculator to find the velocity of a particle in a.
SOLVEDCalculate The Root Mean Square Velocities Of CH4(g), 44 OFF
How do i calculate root mean square velocity? Why is root mean square velocity important in thermodynamics and. The root mean square velocity (rms velocity) is a way to find a single velocity value for the particles. Vᵣₘₛ = √(3 * r * t / m), where r is the gas constant, t is the temperature, and m is the.
Free (RMS) Root mean Square velocity Animation by Joana Barreira
This online chemistry calculator may be used to calculate root mean square velocity, mean velocity (a.k.a average velocity) and median velocity. The root mean square velocity (rms velocity) is a way to find a single velocity value for the particles. Use the root mean square velocity calculator to find the velocity of a particle in a gas based on the.
Solved Calculate the root mean square velocity and
Μ rms = (3rt/m) ½ This online chemistry calculator may be used to calculate root mean square velocity, mean velocity (a.k.a average velocity) and median velocity. Woosh, your result is instantly computed! Vᵣₘₛ = √(3 * r * t / m), where r is the gas constant, t is the temperature, and m is the molar mass. Our root mean.
Woosh, Your Result Is Instantly Computed!
The average velocity of gas particles is found using the root mean square velocity formula: Why is root mean square velocity important in thermodynamics and. How do i calculate root mean square velocity? Vᵣₘₛ = √(3 * r * t / m), where r is the gas constant, t is the temperature, and m is the molar mass.
This Online Chemistry Calculator May Be Used To Calculate Root Mean Square Velocity, Mean Velocity (A.k.a Average Velocity) And Median Velocity.
Use the root mean square velocity calculator to find the velocity of a particle in a gas based on the kinetic theory of gases. Our root mean square speed calculator gives you an effortless way to calculate the rms speed for an ideal and mostly monoatomic gases. The root mean square velocity (rms velocity) is a way to find a single velocity value for the particles. To calculate, we need to: