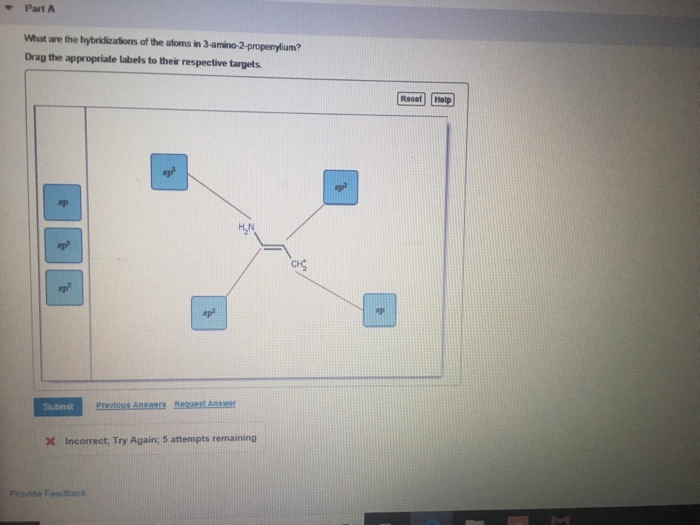
What Are The Hybridizations Of The Atoms In 3 Amino 2 Propenylium
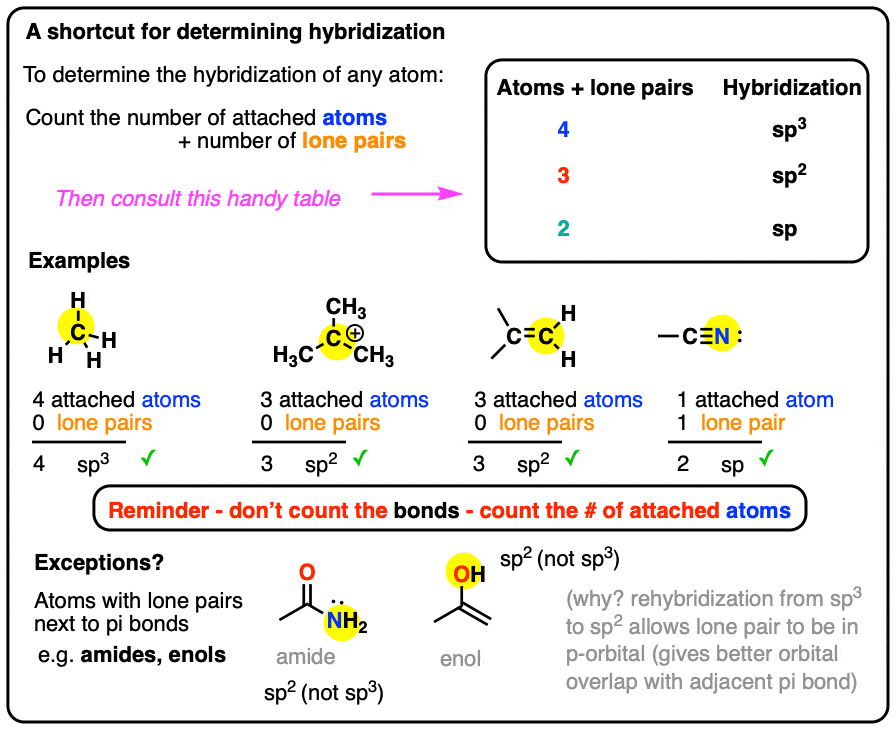
What Are The Hybridizations Of The Atoms In 3 Amino 2 Propenylium - There are 2 steps to solve this one. So, the 3 groups around the central atom gives you the sp 3. 8 rows the shapes of organic molecules may be understood by looking at the hybridization adopted by each of the atoms in the molecule. Drag the appropriate labels to. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. To put it plain, i can summarize the hybridizations in the following picture: If there are 2 regions of electron density, the atom is sp hybridized. If there are 3 regions of electron density, the atom is sp2 hybridized.
Drag the appropriate labels to. If there are 3 regions of electron density, the atom is sp2 hybridized. 8 rows the shapes of organic molecules may be understood by looking at the hybridization adopted by each of the atoms in the molecule. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. There are 2 steps to solve this one. If there are 2 regions of electron density, the atom is sp hybridized. So, the 3 groups around the central atom gives you the sp 3. To put it plain, i can summarize the hybridizations in the following picture:
8 rows the shapes of organic molecules may be understood by looking at the hybridization adopted by each of the atoms in the molecule. Drag the appropriate labels to. If there are 2 regions of electron density, the atom is sp hybridized. So, the 3 groups around the central atom gives you the sp 3. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. To put it plain, i can summarize the hybridizations in the following picture: If there are 3 regions of electron density, the atom is sp2 hybridized. There are 2 steps to solve this one.
میانبر استاد شیمی آلی تولیدی فرمیک
So, the 3 groups around the central atom gives you the sp 3. 8 rows the shapes of organic molecules may be understood by looking at the hybridization adopted by each of the atoms in the molecule. Drag the appropriate labels to. If there are 3 regions of electron density, the atom is sp2 hybridized. There are 2 steps to.
Solved I posted this question once before and they got it
8 rows the shapes of organic molecules may be understood by looking at the hybridization adopted by each of the atoms in the molecule. There are 2 steps to solve this one. Drag the appropriate labels to. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. So, the 3 groups around the central.
Types of Hybridization Definitions, Examples, Key Features, Steps to
If there are 2 regions of electron density, the atom is sp hybridized. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. If there are 3 regions of electron density, the atom is sp2 hybridized. So, the 3 groups around the central atom gives you the sp 3. There are 2 steps to.
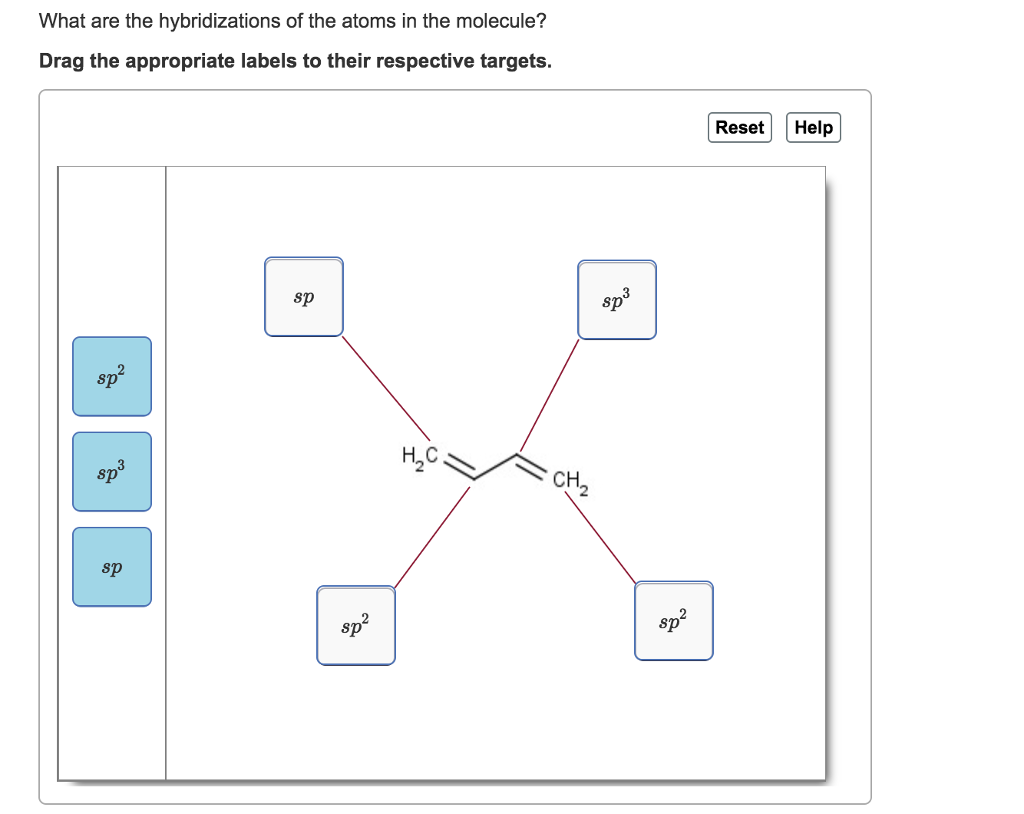
Solved What are the hybridizations of the atoms in the
Drag the appropriate labels to. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. To put it plain, i can summarize the hybridizations in the following picture: So, the 3 groups around the central atom gives you the sp 3. There are 2 steps to solve this one.
What is Hybridization? sp3, sp2, Examples and Formula
8 rows the shapes of organic molecules may be understood by looking at the hybridization adopted by each of the atoms in the molecule. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. Drag the appropriate labels to. There are 2 steps to solve this one. So, the 3 groups around the central.
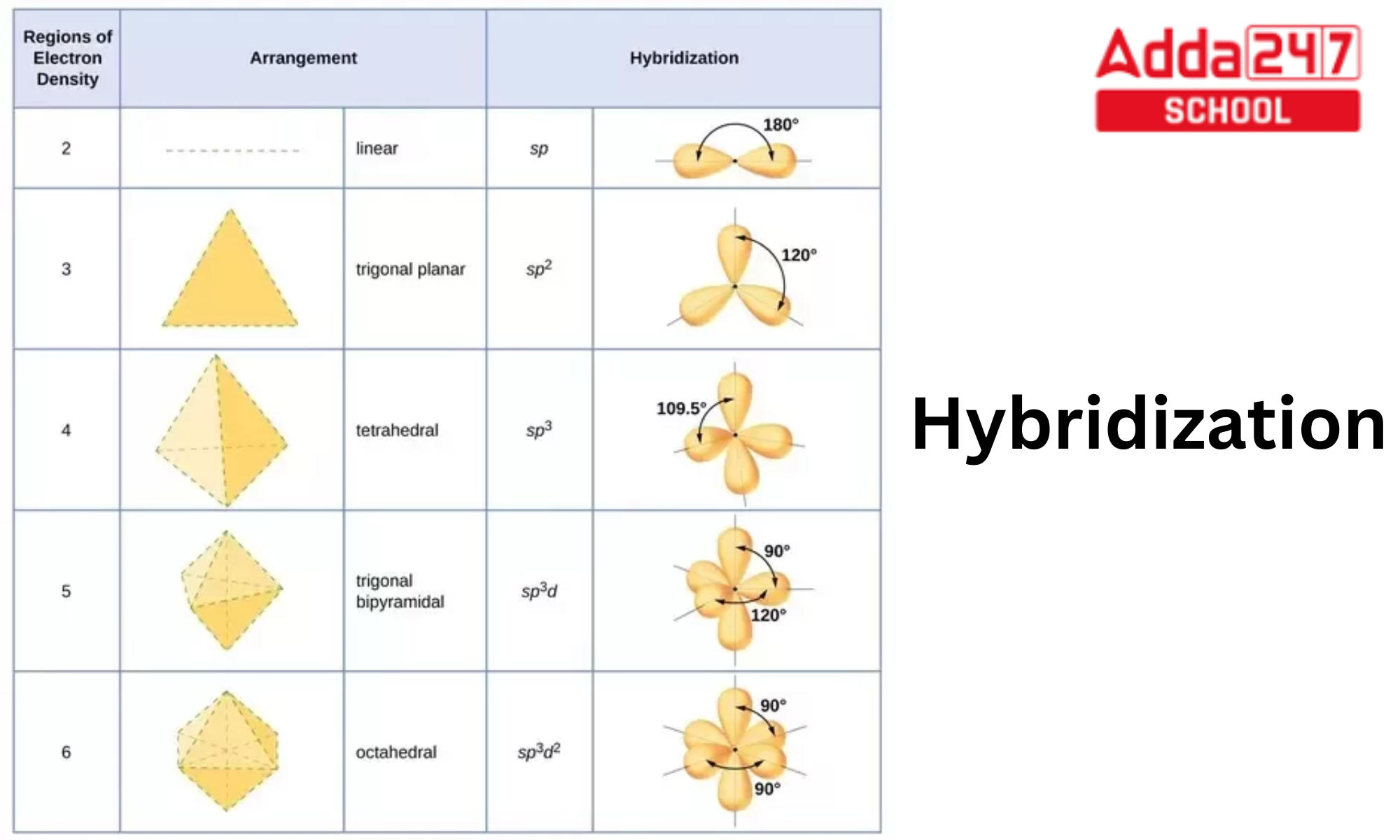
Hybridization Orbitals Chart
You'll get a detailed solution from a subject matter expert that helps you learn core concepts. To put it plain, i can summarize the hybridizations in the following picture: 8 rows the shapes of organic molecules may be understood by looking at the hybridization adopted by each of the atoms in the molecule. So, the 3 groups around the central.
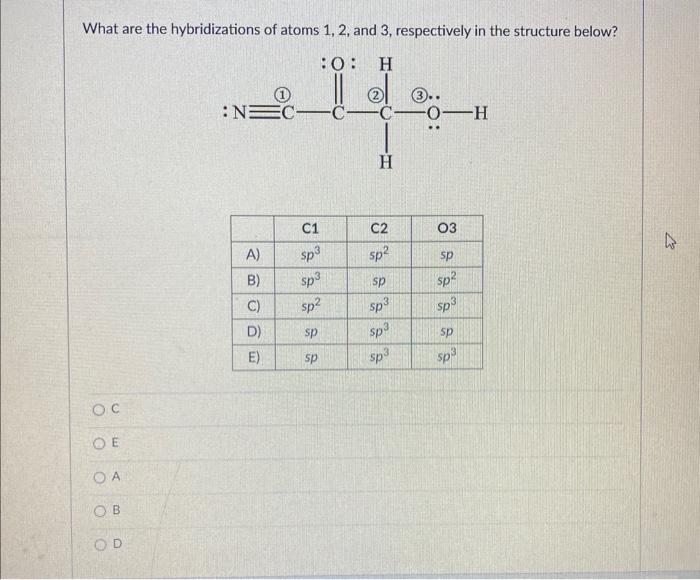
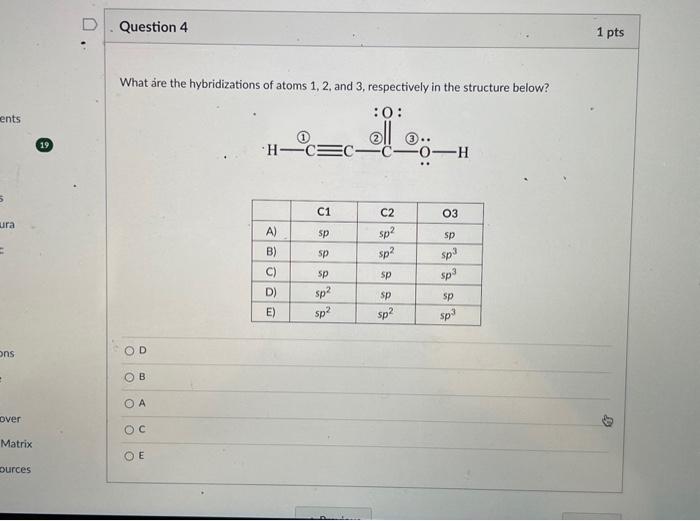
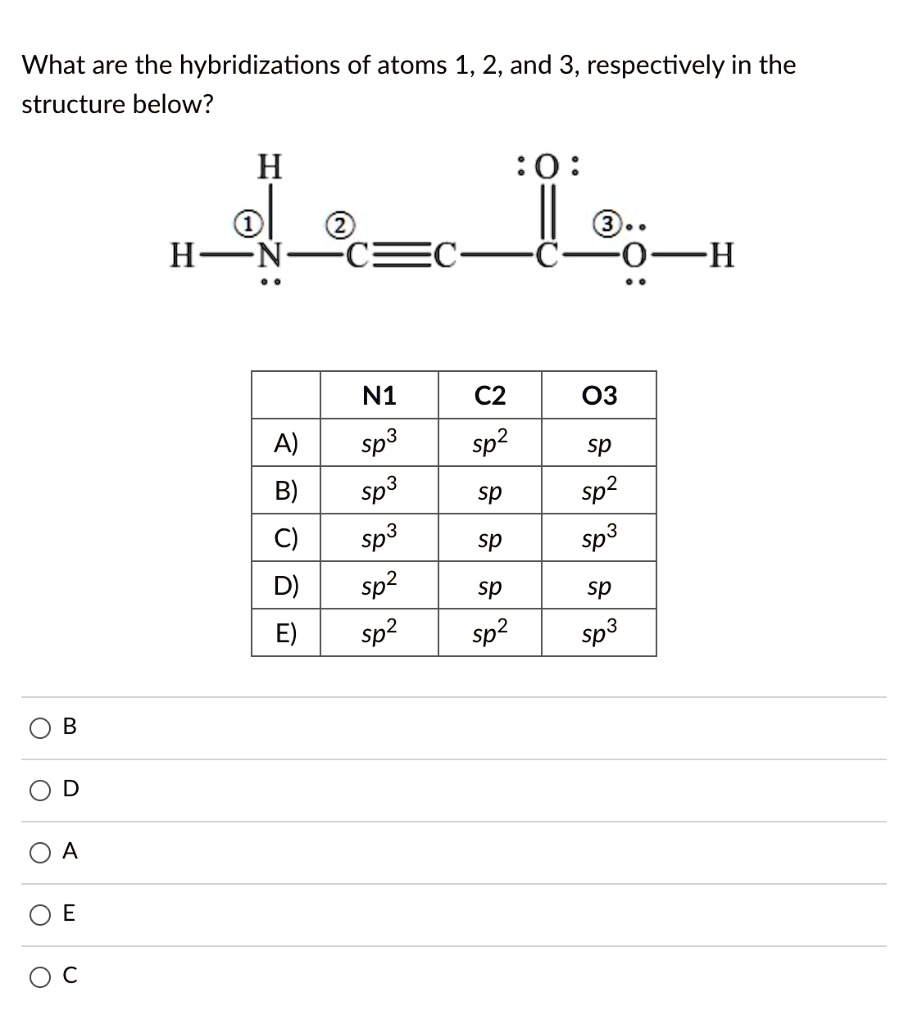
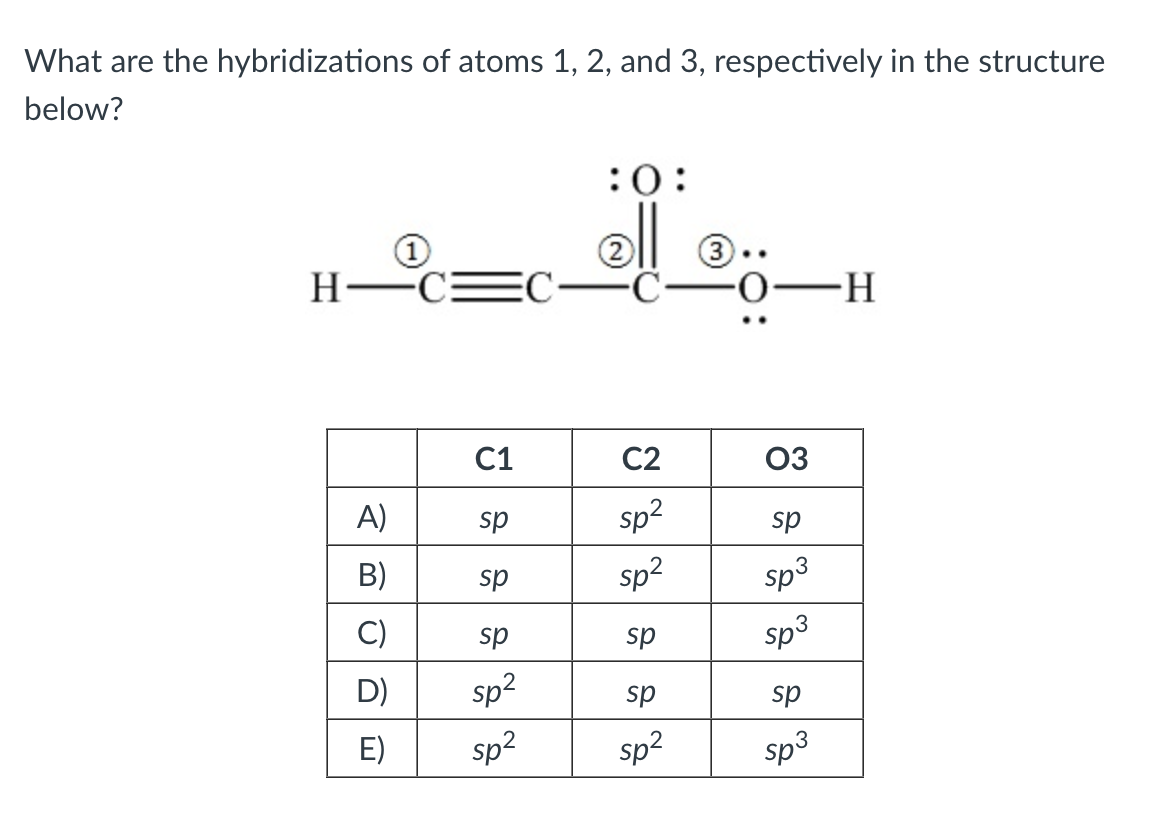
Solved What are the hybridizations of atoms 1,2 , and 3 ,
If there are 2 regions of electron density, the atom is sp hybridized. There are 2 steps to solve this one. Drag the appropriate labels to. 8 rows the shapes of organic molecules may be understood by looking at the hybridization adopted by each of the atoms in the molecule. To put it plain, i can summarize the hybridizations in.
Solved What are the hybridizations of atoms 1,2 , and 3 ,
You'll get a detailed solution from a subject matter expert that helps you learn core concepts. If there are 2 regions of electron density, the atom is sp hybridized. Drag the appropriate labels to. To put it plain, i can summarize the hybridizations in the following picture: There are 2 steps to solve this one.
what are the hybridizations of atoms 1 2and 3 respectively in the
To put it plain, i can summarize the hybridizations in the following picture: So, the 3 groups around the central atom gives you the sp 3. There are 2 steps to solve this one. If there are 3 regions of electron density, the atom is sp2 hybridized. If there are 2 regions of electron density, the atom is sp hybridized.
Solved What are the hybridizations of atoms 1,2 , and 3 ,
Drag the appropriate labels to. 8 rows the shapes of organic molecules may be understood by looking at the hybridization adopted by each of the atoms in the molecule. If there are 3 regions of electron density, the atom is sp2 hybridized. To put it plain, i can summarize the hybridizations in the following picture: If there are 2 regions.
So, The 3 Groups Around The Central Atom Gives You The Sp 3.
To put it plain, i can summarize the hybridizations in the following picture: If there are 2 regions of electron density, the atom is sp hybridized. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. Drag the appropriate labels to.
If There Are 3 Regions Of Electron Density, The Atom Is Sp2 Hybridized.
8 rows the shapes of organic molecules may be understood by looking at the hybridization adopted by each of the atoms in the molecule. There are 2 steps to solve this one.