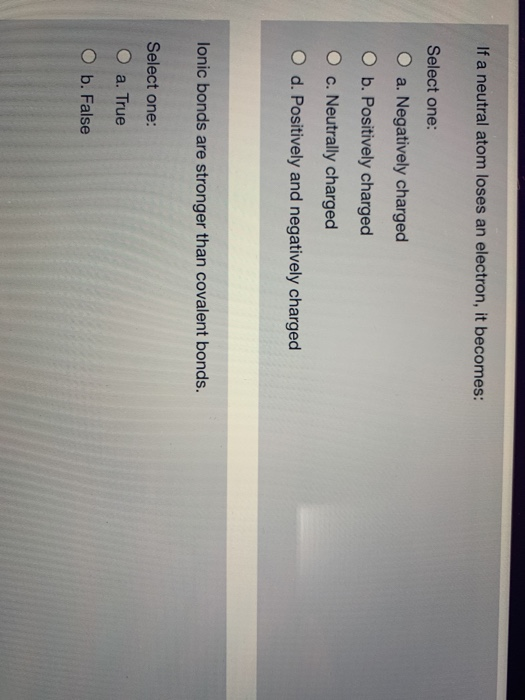
What Happens If An Atom Loses An Electron
What Happens If An Atom Loses An Electron - Atoms that lose electrons acquire a positive charge because they are left with fewer negatively charged electrons to balance the. When an atom loses or gains an electron, it undergoes a process called ionization, resulting in the formation of an ion. This is because the atom now has more. When an atom loses an electron from its outermost shell, it becomes a positive ion. Let's break down why this happens: When an atom loses an electron, it undergoes a change in charge.
Let's break down why this happens: This is because the atom now has more. Atoms that lose electrons acquire a positive charge because they are left with fewer negatively charged electrons to balance the. When an atom loses an electron from its outermost shell, it becomes a positive ion. When an atom loses or gains an electron, it undergoes a process called ionization, resulting in the formation of an ion. When an atom loses an electron, it undergoes a change in charge.
When an atom loses an electron from its outermost shell, it becomes a positive ion. When an atom loses an electron, it undergoes a change in charge. This is because the atom now has more. Let's break down why this happens: When an atom loses or gains an electron, it undergoes a process called ionization, resulting in the formation of an ion. Atoms that lose electrons acquire a positive charge because they are left with fewer negatively charged electrons to balance the.
Solved If a neutral atom loses an electron, it
Atoms that lose electrons acquire a positive charge because they are left with fewer negatively charged electrons to balance the. When an atom loses an electron, it undergoes a change in charge. When an atom loses or gains an electron, it undergoes a process called ionization, resulting in the formation of an ion. This is because the atom now has.
Solved If an atom loses an electron, it is (a) negatively
When an atom loses an electron, it undergoes a change in charge. Let's break down why this happens: Atoms that lose electrons acquire a positive charge because they are left with fewer negatively charged electrons to balance the. When an atom loses or gains an electron, it undergoes a process called ionization, resulting in the formation of an ion. This.
16 What happens when an atom gains or loses electrons 16 What
Atoms that lose electrons acquire a positive charge because they are left with fewer negatively charged electrons to balance the. When an atom loses an electron, it undergoes a change in charge. When an atom loses an electron from its outermost shell, it becomes a positive ion. When an atom loses or gains an electron, it undergoes a process called.
SOLVED What happens to an atom of sulfur (S) if it loses an electron
Let's break down why this happens: When an atom loses or gains an electron, it undergoes a process called ionization, resulting in the formation of an ion. Atoms that lose electrons acquire a positive charge because they are left with fewer negatively charged electrons to balance the. When an atom loses an electron from its outermost shell, it becomes a.
When an atom loses an electron, it MakeTheBrainHappy
Atoms that lose electrons acquire a positive charge because they are left with fewer negatively charged electrons to balance the. When an atom loses an electron, it undergoes a change in charge. When an atom loses or gains an electron, it undergoes a process called ionization, resulting in the formation of an ion. When an atom loses an electron from.
WHAT HAPPENS IF YOU SPLIT AN ATOM? LikeFigures
This is because the atom now has more. When an atom loses an electron, it undergoes a change in charge. Let's break down why this happens: Atoms that lose electrons acquire a positive charge because they are left with fewer negatively charged electrons to balance the. When an atom loses an electron from its outermost shell, it becomes a positive.
an atom loses an electron. he says i really gotta keep an ion them
Let's break down why this happens: When an atom loses an electron from its outermost shell, it becomes a positive ion. Atoms that lose electrons acquire a positive charge because they are left with fewer negatively charged electrons to balance the. This is because the atom now has more. When an atom loses an electron, it undergoes a change in.
WHAT HAPPENS IF YOU SPLIT AN ATOM mousetimes
When an atom loses an electron from its outermost shell, it becomes a positive ion. Let's break down why this happens: This is because the atom now has more. When an atom loses or gains an electron, it undergoes a process called ionization, resulting in the formation of an ion. When an atom loses an electron, it undergoes a change.
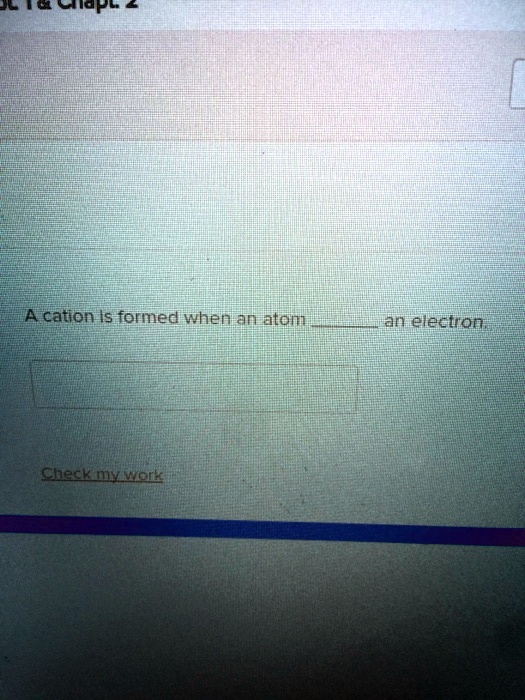
SOLVED A cation is formed when an atom loses an electron.
When an atom loses an electron, it undergoes a change in charge. When an atom loses or gains an electron, it undergoes a process called ionization, resulting in the formation of an ion. When an atom loses an electron from its outermost shell, it becomes a positive ion. Atoms that lose electrons acquire a positive charge because they are left.
When an Atom Loses an Electron What Is Its Charge What is the charge
When an atom loses or gains an electron, it undergoes a process called ionization, resulting in the formation of an ion. Let's break down why this happens: When an atom loses an electron, it undergoes a change in charge. When an atom loses an electron from its outermost shell, it becomes a positive ion. Atoms that lose electrons acquire a.
This Is Because The Atom Now Has More.
Atoms that lose electrons acquire a positive charge because they are left with fewer negatively charged electrons to balance the. When an atom loses an electron, it undergoes a change in charge. When an atom loses an electron from its outermost shell, it becomes a positive ion. Let's break down why this happens: