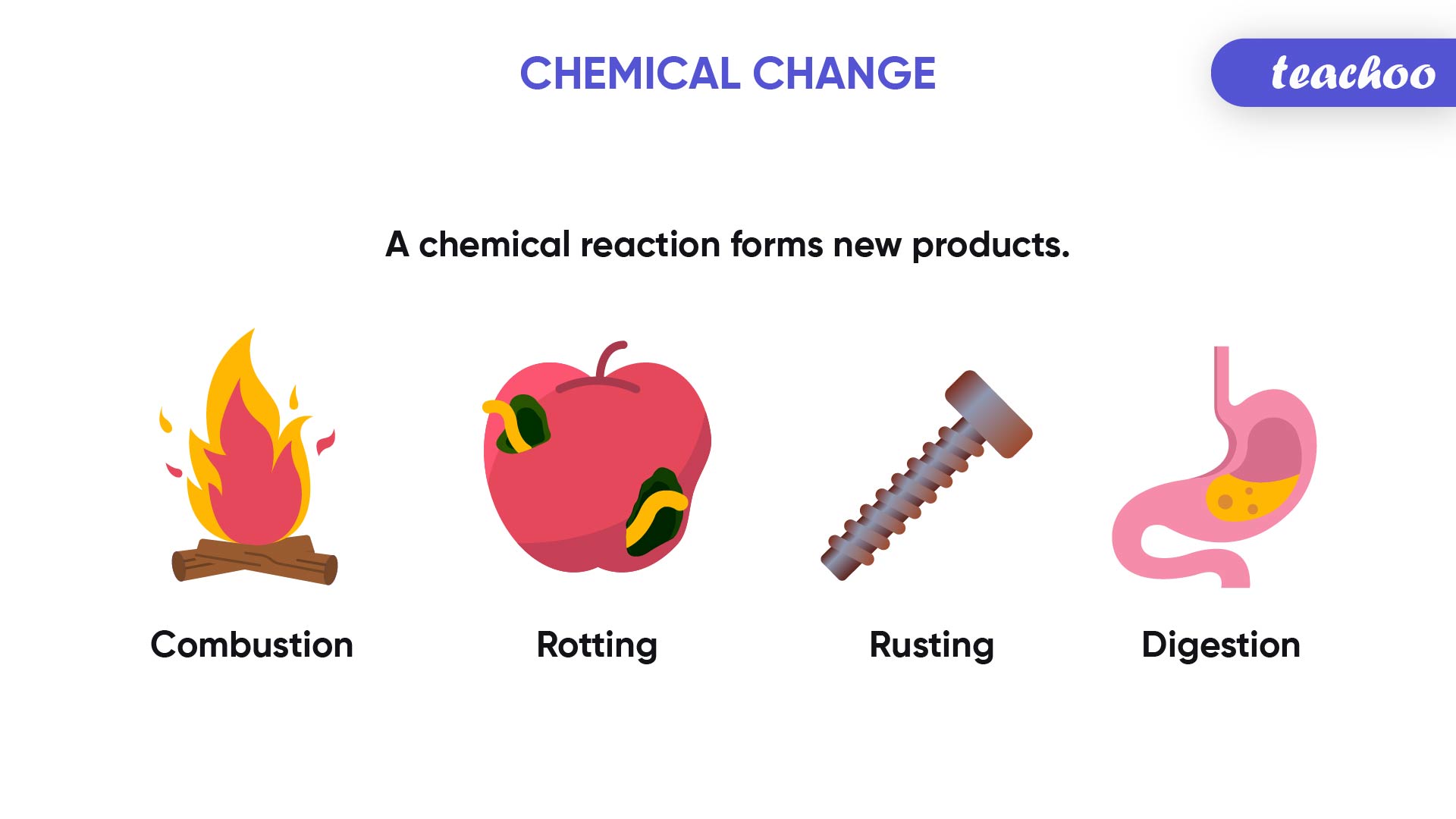
What Is Always Conserved In A Chemical Reaction
What Is Always Conserved In A Chemical Reaction - We can predict the masses of products and reactants involved in chemical reactions as. During a chemical reaction no atoms are created or destroyed. Chemical reaction when chemical bonds are broken and made between atoms, so that new substances (compounds or elements). Why is mass always conserved in a chemical reaction? In every chemical reaction, the same mass of matter must end up in the products as started in the reactants. This means that there are. Mass is conserved in chemical reactions. This means that the mass of substances present at the start of a reaction (reactants) must be equal to the mass of those.
In every chemical reaction, the same mass of matter must end up in the products as started in the reactants. During a chemical reaction no atoms are created or destroyed. Chemical reaction when chemical bonds are broken and made between atoms, so that new substances (compounds or elements). We can predict the masses of products and reactants involved in chemical reactions as. Why is mass always conserved in a chemical reaction? This means that there are. This means that the mass of substances present at the start of a reaction (reactants) must be equal to the mass of those. Mass is conserved in chemical reactions.
During a chemical reaction no atoms are created or destroyed. This means that the mass of substances present at the start of a reaction (reactants) must be equal to the mass of those. In every chemical reaction, the same mass of matter must end up in the products as started in the reactants. Chemical reaction when chemical bonds are broken and made between atoms, so that new substances (compounds or elements). Why is mass always conserved in a chemical reaction? Mass is conserved in chemical reactions. We can predict the masses of products and reactants involved in chemical reactions as. This means that there are.
SOLVED Which of the following are CONSERVED in every chemical reaction
Why is mass always conserved in a chemical reaction? Mass is conserved in chemical reactions. This means that there are. During a chemical reaction no atoms are created or destroyed. Chemical reaction when chemical bonds are broken and made between atoms, so that new substances (compounds or elements).
What Is A Chemical Reaction
This means that the mass of substances present at the start of a reaction (reactants) must be equal to the mass of those. In every chemical reaction, the same mass of matter must end up in the products as started in the reactants. Mass is conserved in chemical reactions. We can predict the masses of products and reactants involved in.
Trendy Chemical Reaction 35887296 Vector Art at Vecteezy
Chemical reaction when chemical bonds are broken and made between atoms, so that new substances (compounds or elements). Why is mass always conserved in a chemical reaction? We can predict the masses of products and reactants involved in chemical reactions as. This means that the mass of substances present at the start of a reaction (reactants) must be equal to.
Chemical Reaction Definition, Types and Examples Class 10 Science
Why is mass always conserved in a chemical reaction? This means that there are. This means that the mass of substances present at the start of a reaction (reactants) must be equal to the mass of those. We can predict the masses of products and reactants involved in chemical reactions as. Chemical reaction when chemical bonds are broken and made.
[Solved] Is mass or number of moles always conserved in a chemical
This means that the mass of substances present at the start of a reaction (reactants) must be equal to the mass of those. Mass is conserved in chemical reactions. This means that there are. Chemical reaction when chemical bonds are broken and made between atoms, so that new substances (compounds or elements). We can predict the masses of products and.
Which of these is/are conserved during a chemical reaction? Filo
We can predict the masses of products and reactants involved in chemical reactions as. This means that there are. In every chemical reaction, the same mass of matter must end up in the products as started in the reactants. Why is mass always conserved in a chemical reaction? Mass is conserved in chemical reactions.
Solved 1. Which of the following is/are conserved in a
We can predict the masses of products and reactants involved in chemical reactions as. This means that the mass of substances present at the start of a reaction (reactants) must be equal to the mass of those. This means that there are. Chemical reaction when chemical bonds are broken and made between atoms, so that new substances (compounds or elements)..
What Is Conserved in Chemical Reactions? Sciencing
Why is mass always conserved in a chemical reaction? Mass is conserved in chemical reactions. In every chemical reaction, the same mass of matter must end up in the products as started in the reactants. This means that there are. During a chemical reaction no atoms are created or destroyed.
Solved Which of the following is not always conserved in a
We can predict the masses of products and reactants involved in chemical reactions as. In every chemical reaction, the same mass of matter must end up in the products as started in the reactants. This means that the mass of substances present at the start of a reaction (reactants) must be equal to the mass of those. This means that.
Energy Is Conserved Always Home Page
This means that the mass of substances present at the start of a reaction (reactants) must be equal to the mass of those. Mass is conserved in chemical reactions. In every chemical reaction, the same mass of matter must end up in the products as started in the reactants. We can predict the masses of products and reactants involved in.
This Means That The Mass Of Substances Present At The Start Of A Reaction (Reactants) Must Be Equal To The Mass Of Those.
This means that there are. Chemical reaction when chemical bonds are broken and made between atoms, so that new substances (compounds or elements). Why is mass always conserved in a chemical reaction? During a chemical reaction no atoms are created or destroyed.
We Can Predict The Masses Of Products And Reactants Involved In Chemical Reactions As.
In every chemical reaction, the same mass of matter must end up in the products as started in the reactants. Mass is conserved in chemical reactions.