What Is Oxidation Number Of Chromium In Crso4
What Is Oxidation Number Of Chromium In Crso4 - In cr2 (so4)3, the so4 ion has a. Not the question you’re looking for? Here’s the best way to solve it. Oxidation state is the number that. To find the oxidation number of chromium in crso4, we need to consider the oxidation states of the other atoms in the compound. To maintain neutrality, cr must have an oxidation number of +2. In the compound crso₄, the oxidation number of chr. Since cr c r is in column 3 3 of the periodic table, it will share 3 3 electrons and use an oxidation state of 3 3. Since o o is in column 6 6 of the. The oxidation state of chromium in the given compound is +4.
Since o o is in column 6 6 of the. In cr2 (so4)3, the so4 ion has a. To find the oxidation number of chromium in crso4, we need to consider the oxidation states of the other atoms in the compound. Since cr c r is in column 3 3 of the periodic table, it will share 3 3 electrons and use an oxidation state of 3 3. In the compound crso₄, the oxidation number of chr. Not the question you’re looking for? Here’s the best way to solve it. To maintain neutrality, cr must have an oxidation number of +2. Oxidation state is the number that. The oxidation state of chromium in the given compound is +4.
The oxidation state of chromium in the given compound is +4. In cr2 (so4)3, the so4 ion has a. Here’s the best way to solve it. To maintain neutrality, cr must have an oxidation number of +2. Since cr c r is in column 3 3 of the periodic table, it will share 3 3 electrons and use an oxidation state of 3 3. Since o o is in column 6 6 of the. Not the question you’re looking for? Oxidation state is the number that. In the compound crso₄, the oxidation number of chr. To find the oxidation number of chromium in crso4, we need to consider the oxidation states of the other atoms in the compound.
Oxidation Number of Chromium
Oxidation state is the number that. To maintain neutrality, cr must have an oxidation number of +2. In cr2 (so4)3, the so4 ion has a. To find the oxidation number of chromium in crso4, we need to consider the oxidation states of the other atoms in the compound. Here’s the best way to solve it.
What is the oxidation number of chromium of this substance Cr2O^27
Here’s the best way to solve it. In the compound crso₄, the oxidation number of chr. In cr2 (so4)3, the so4 ion has a. To find the oxidation number of chromium in crso4, we need to consider the oxidation states of the other atoms in the compound. Since cr c r is in column 3 3 of the periodic table,.
Give the Oxidation Number of Chromium in the Following AlinahasSherman
Not the question you’re looking for? To find the oxidation number of chromium in crso4, we need to consider the oxidation states of the other atoms in the compound. In cr2 (so4)3, the so4 ion has a. Here’s the best way to solve it. Since cr c r is in column 3 3 of the periodic table, it will share.
The oxidation number of chromium in potassium dichromate is 11. The oxida..
The oxidation state of chromium in the given compound is +4. In the compound crso₄, the oxidation number of chr. Since cr c r is in column 3 3 of the periodic table, it will share 3 3 electrons and use an oxidation state of 3 3. To find the oxidation number of chromium in crso4, we need to consider.
The oxidation number of chromium in K2 Cr2 O, is Filo
To maintain neutrality, cr must have an oxidation number of +2. In cr2 (so4)3, the so4 ion has a. Oxidation state is the number that. Since o o is in column 6 6 of the. In the compound crso₄, the oxidation number of chr.
What is the oxidation number of chromium in K3CrO8 Please explain its
To find the oxidation number of chromium in crso4, we need to consider the oxidation states of the other atoms in the compound. In the compound crso₄, the oxidation number of chr. To maintain neutrality, cr must have an oxidation number of +2. Since o o is in column 6 6 of the. Here’s the best way to solve it.
The oxidation number of chromium in CrO5 is
In cr2 (so4)3, the so4 ion has a. Since cr c r is in column 3 3 of the periodic table, it will share 3 3 electrons and use an oxidation state of 3 3. The oxidation state of chromium in the given compound is +4. In the compound crso₄, the oxidation number of chr. To maintain neutrality, cr must.
Give the Oxidation Number of Chromium in the Following AlinahasSherman
In the compound crso₄, the oxidation number of chr. The oxidation state of chromium in the given compound is +4. To maintain neutrality, cr must have an oxidation number of +2. Not the question you’re looking for? In cr2 (so4)3, the so4 ion has a.
SOLVED What is the oxidation number of chromium in K2Cr2O7
To maintain neutrality, cr must have an oxidation number of +2. The oxidation state of chromium in the given compound is +4. Since cr c r is in column 3 3 of the periodic table, it will share 3 3 electrons and use an oxidation state of 3 3. Since o o is in column 6 6 of the. To.
Oxidation number of chromium in [cr(nh3)6][Co(cn)6] Chemistry
To maintain neutrality, cr must have an oxidation number of +2. Not the question you’re looking for? To find the oxidation number of chromium in crso4, we need to consider the oxidation states of the other atoms in the compound. In the compound crso₄, the oxidation number of chr. The oxidation state of chromium in the given compound is +4.
The Oxidation State Of Chromium In The Given Compound Is +4.
To maintain neutrality, cr must have an oxidation number of +2. Here’s the best way to solve it. In cr2 (so4)3, the so4 ion has a. To find the oxidation number of chromium in crso4, we need to consider the oxidation states of the other atoms in the compound.
Not The Question You’re Looking For?
In the compound crso₄, the oxidation number of chr. Since o o is in column 6 6 of the. Since cr c r is in column 3 3 of the periodic table, it will share 3 3 electrons and use an oxidation state of 3 3. Oxidation state is the number that.







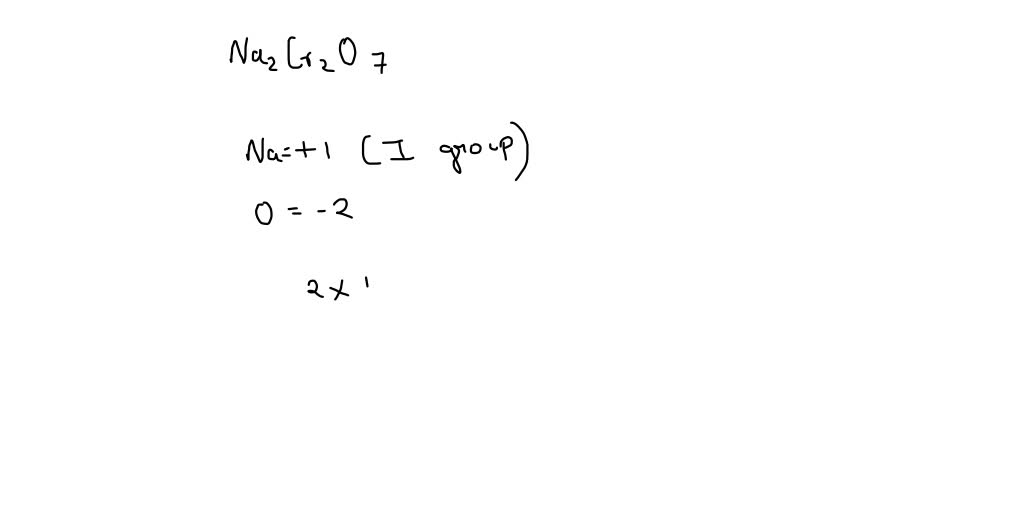
![Oxidation number of chromium in [cr(nh3)6][Co(cn)6] Chemistry](https://s3mn.mnimgs.com/img/shared/content_ck_images/ck_5c72e54905d74.jpg)