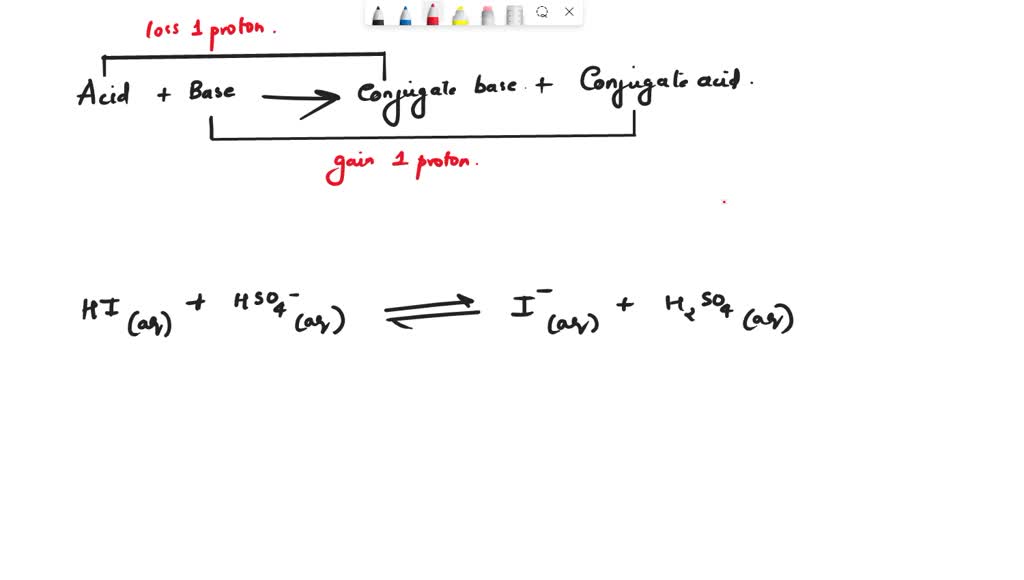What Is The Conjugate Base Of H2So4
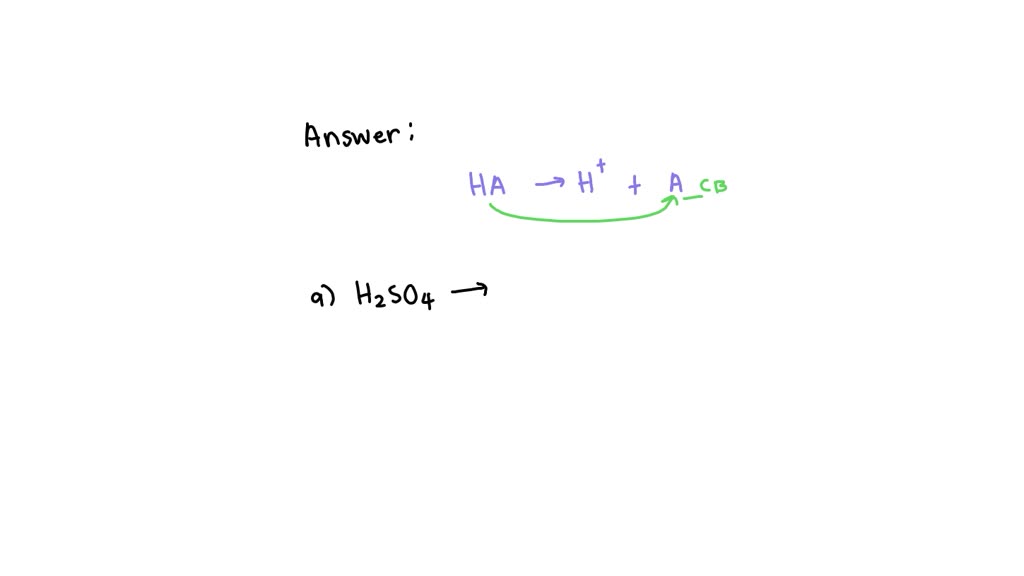
What Is The Conjugate Base Of H2So4 - There are 2 steps to solve this one. The conjugate base of a strong acid is a very weak base, and, conversely, the conjugate acid of a strong base is a very weak. The chemical reaction for the above. The conjugate base of h 2 s o 4 in the following reaction is: The conjugate base of a substance is formed by removing a proton (h a +) from it. Sulfuric acid (h2so4) will lose a proton to form its conjugate base. H 2 s o 4 + h 2 o → h 3 o + + h s o − 4 To find the conjugate base of an acid, you remove a proton (h+) from it.
The chemical reaction for the above. There are 2 steps to solve this one. Sulfuric acid (h2so4) will lose a proton to form its conjugate base. H 2 s o 4 + h 2 o → h 3 o + + h s o − 4 The conjugate base of a strong acid is a very weak base, and, conversely, the conjugate acid of a strong base is a very weak. The conjugate base of h 2 s o 4 in the following reaction is: The conjugate base of a substance is formed by removing a proton (h a +) from it. To find the conjugate base of an acid, you remove a proton (h+) from it.
The conjugate base of a strong acid is a very weak base, and, conversely, the conjugate acid of a strong base is a very weak. There are 2 steps to solve this one. To find the conjugate base of an acid, you remove a proton (h+) from it. The conjugate base of a substance is formed by removing a proton (h a +) from it. H 2 s o 4 + h 2 o → h 3 o + + h s o − 4 The conjugate base of h 2 s o 4 in the following reaction is: The chemical reaction for the above. Sulfuric acid (h2so4) will lose a proton to form its conjugate base.
SOLVED Identify the conjugate base for each acid. * conjugate base of
To find the conjugate base of an acid, you remove a proton (h+) from it. The conjugate base of h 2 s o 4 in the following reaction is: Sulfuric acid (h2so4) will lose a proton to form its conjugate base. The conjugate base of a substance is formed by removing a proton (h a +) from it. H 2.
SOLUTION Conjugate Acid and Conjugate Base pKa Chart Studypool
H 2 s o 4 + h 2 o → h 3 o + + h s o − 4 There are 2 steps to solve this one. The chemical reaction for the above. The conjugate base of h 2 s o 4 in the following reaction is: The conjugate base of a substance is formed by removing a proton.
SOLVED Texts dropdown menu 1. conjugate acid or conjugate base 2
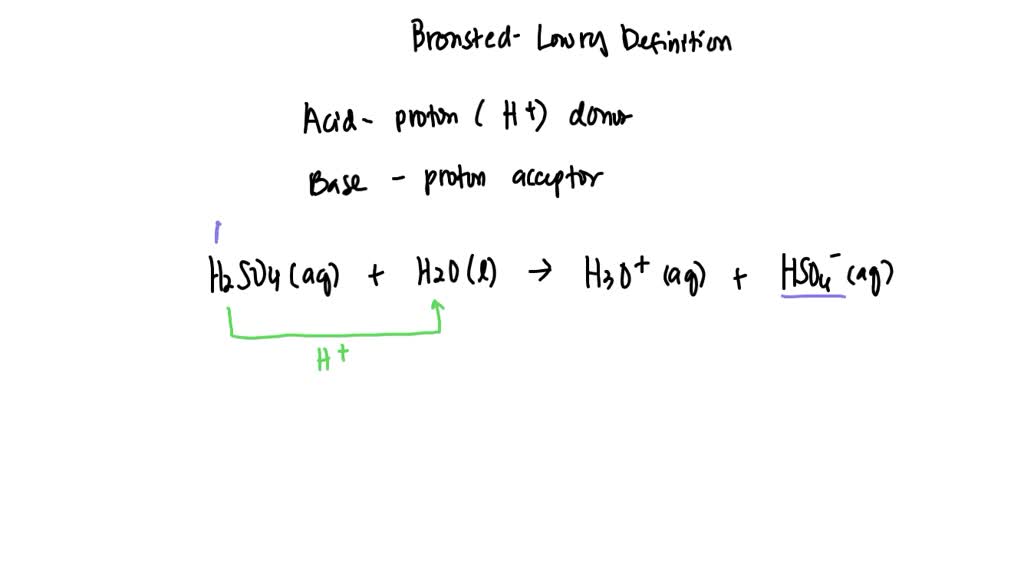
H 2 s o 4 + h 2 o → h 3 o + + h s o − 4 Sulfuric acid (h2so4) will lose a proton to form its conjugate base. The conjugate base of h 2 s o 4 in the following reaction is: The conjugate base of a substance is formed by removing a proton (h a.
SOLVED Identify the conjugate base for each acid. conjugate base of
Sulfuric acid (h2so4) will lose a proton to form its conjugate base. The conjugate base of a strong acid is a very weak base, and, conversely, the conjugate acid of a strong base is a very weak. The conjugate base of h 2 s o 4 in the following reaction is: There are 2 steps to solve this one. The.
Solved Draw the conjugate base of the following Acids
H 2 s o 4 + h 2 o → h 3 o + + h s o − 4 The conjugate base of h 2 s o 4 in the following reaction is: There are 2 steps to solve this one. The conjugate base of a strong acid is a very weak base, and, conversely, the conjugate acid of.
H2SO4(aq)+H2O(l)←→H3O+(aq)+HSO4−(aq) acid H2SO4 conjugate base HSO4−
The conjugate base of a strong acid is a very weak base, and, conversely, the conjugate acid of a strong base is a very weak. The conjugate base of h 2 s o 4 in the following reaction is: Sulfuric acid (h2so4) will lose a proton to form its conjugate base. H 2 s o 4 + h 2 o.
Solved Give The Conjugate Base For Each Compound Below. A...
The conjugate base of a strong acid is a very weak base, and, conversely, the conjugate acid of a strong base is a very weak. The conjugate base of h 2 s o 4 in the following reaction is: Sulfuric acid (h2so4) will lose a proton to form its conjugate base. To find the conjugate base of an acid, you.
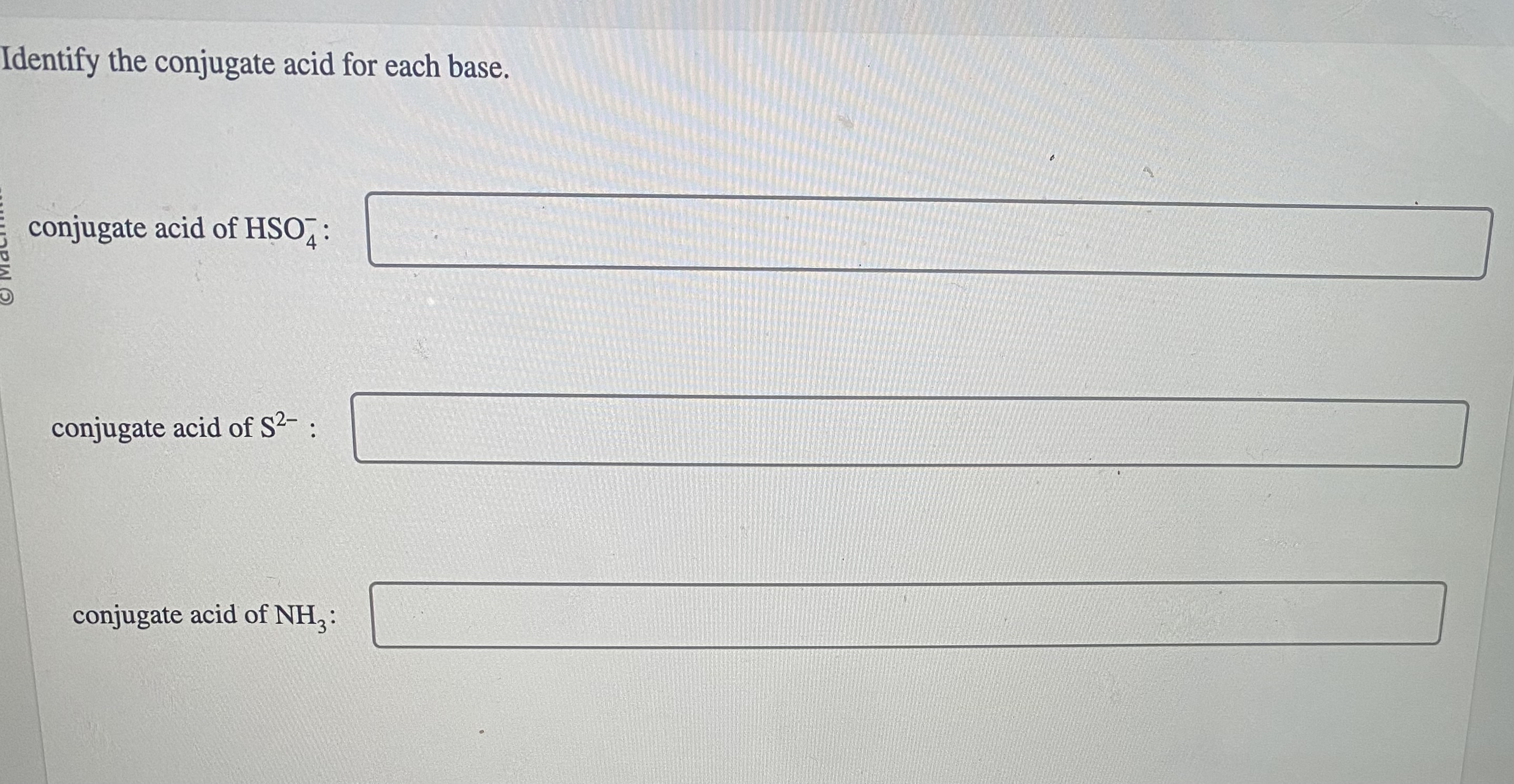
Solved Identify the conjugate acid for each base. conjugate
The conjugate base of a strong acid is a very weak base, and, conversely, the conjugate acid of a strong base is a very weak. H 2 s o 4 + h 2 o → h 3 o + + h s o − 4 The conjugate base of a substance is formed by removing a proton (h a +).
Conjugate Acid Base Chart
Sulfuric acid (h2so4) will lose a proton to form its conjugate base. H 2 s o 4 + h 2 o → h 3 o + + h s o − 4 There are 2 steps to solve this one. To find the conjugate base of an acid, you remove a proton (h+) from it. The conjugate base of a.
SOLUTION Conjugate Acid and Conjugate Base pKa Chart Studypool
The conjugate base of h 2 s o 4 in the following reaction is: Sulfuric acid (h2so4) will lose a proton to form its conjugate base. The conjugate base of a substance is formed by removing a proton (h a +) from it. H 2 s o 4 + h 2 o → h 3 o + + h s.
The Chemical Reaction For The Above.
The conjugate base of a strong acid is a very weak base, and, conversely, the conjugate acid of a strong base is a very weak. The conjugate base of h 2 s o 4 in the following reaction is: H 2 s o 4 + h 2 o → h 3 o + + h s o − 4 There are 2 steps to solve this one.
To Find The Conjugate Base Of An Acid, You Remove A Proton (H+) From It.
The conjugate base of a substance is formed by removing a proton (h a +) from it. Sulfuric acid (h2so4) will lose a proton to form its conjugate base.