What Is The Electron Configuration For The Fe2 Ion
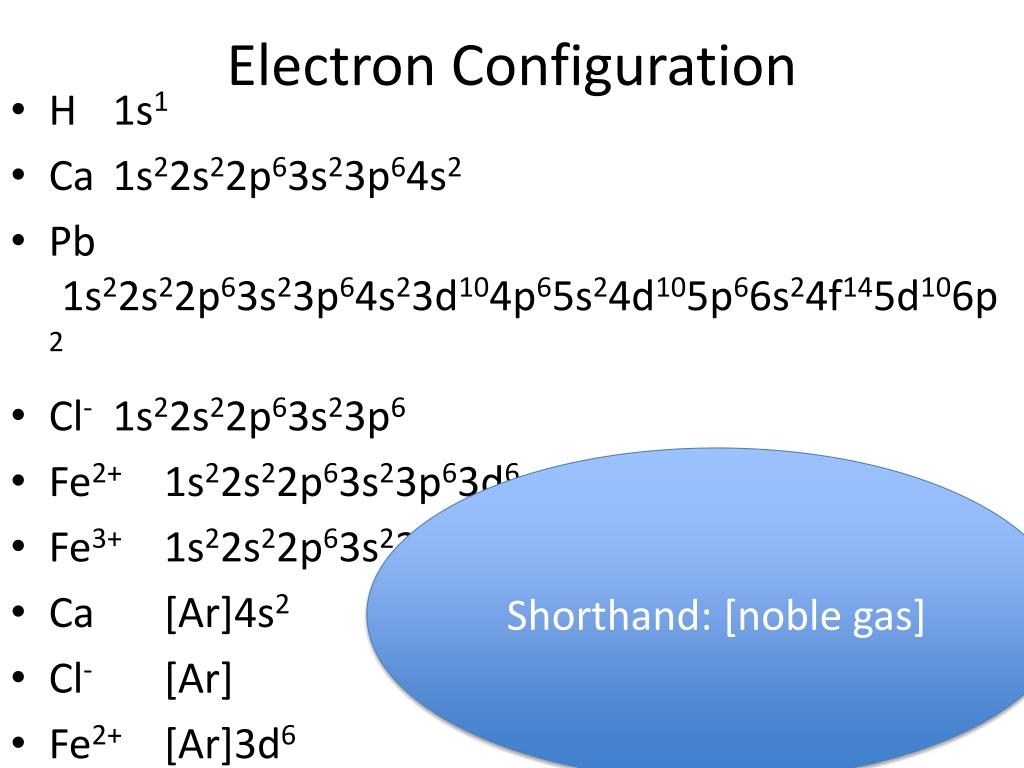
What Is The Electron Configuration For The Fe2 Ion - This means there are 26 electrons in an iron atom, with 2 in the 4s. In order to write the iron electron configuration we first need to know the number of electrons for the fe atom (there are 26 electrons). The electronic configuration of fe is $\ce{[ar] 3d^6 4s^2}$. The electron configuration of iron (fe) is [ar] 3d^6 4s^2. So after removing two electrons the configuration becomes:
This means there are 26 electrons in an iron atom, with 2 in the 4s. In order to write the iron electron configuration we first need to know the number of electrons for the fe atom (there are 26 electrons). The electronic configuration of fe is $\ce{[ar] 3d^6 4s^2}$. The electron configuration of iron (fe) is [ar] 3d^6 4s^2. So after removing two electrons the configuration becomes:
In order to write the iron electron configuration we first need to know the number of electrons for the fe atom (there are 26 electrons). The electron configuration of iron (fe) is [ar] 3d^6 4s^2. This means there are 26 electrons in an iron atom, with 2 in the 4s. The electronic configuration of fe is $\ce{[ar] 3d^6 4s^2}$. So after removing two electrons the configuration becomes:
Which of the following is the stable electronic configuration of Fe^3
The electron configuration of iron (fe) is [ar] 3d^6 4s^2. The electronic configuration of fe is $\ce{[ar] 3d^6 4s^2}$. This means there are 26 electrons in an iron atom, with 2 in the 4s. So after removing two electrons the configuration becomes: In order to write the iron electron configuration we first need to know the number of electrons for.
Electron Configuration Of Ions Worksheet
So after removing two electrons the configuration becomes: This means there are 26 electrons in an iron atom, with 2 in the 4s. The electron configuration of iron (fe) is [ar] 3d^6 4s^2. In order to write the iron electron configuration we first need to know the number of electrons for the fe atom (there are 26 electrons). The electronic.
Electron Configuration For Iron
This means there are 26 electrons in an iron atom, with 2 in the 4s. So after removing two electrons the configuration becomes: In order to write the iron electron configuration we first need to know the number of electrons for the fe atom (there are 26 electrons). The electron configuration of iron (fe) is [ar] 3d^6 4s^2. The electronic.
Which Ion Has the Electron Configuration 1s22s22p63s23p6
In order to write the iron electron configuration we first need to know the number of electrons for the fe atom (there are 26 electrons). This means there are 26 electrons in an iron atom, with 2 in the 4s. So after removing two electrons the configuration becomes: The electron configuration of iron (fe) is [ar] 3d^6 4s^2. The electronic.
Iron Electron Configuration
The electron configuration of iron (fe) is [ar] 3d^6 4s^2. In order to write the iron electron configuration we first need to know the number of electrons for the fe atom (there are 26 electrons). This means there are 26 electrons in an iron atom, with 2 in the 4s. The electronic configuration of fe is $\ce{[ar] 3d^6 4s^2}$. So.
Periodic Table With Charges And Electron Configuration Review Home Decor
So after removing two electrons the configuration becomes: In order to write the iron electron configuration we first need to know the number of electrons for the fe atom (there are 26 electrons). This means there are 26 electrons in an iron atom, with 2 in the 4s. The electronic configuration of fe is $\ce{[ar] 3d^6 4s^2}$. The electron configuration.
Electron Configuration For Fe3+
So after removing two electrons the configuration becomes: The electron configuration of iron (fe) is [ar] 3d^6 4s^2. In order to write the iron electron configuration we first need to know the number of electrons for the fe atom (there are 26 electrons). This means there are 26 electrons in an iron atom, with 2 in the 4s. The electronic.
Electron Configuration for Iron (Fe and Fe2+, Fe3+ ions)
In order to write the iron electron configuration we first need to know the number of electrons for the fe atom (there are 26 electrons). This means there are 26 electrons in an iron atom, with 2 in the 4s. The electron configuration of iron (fe) is [ar] 3d^6 4s^2. So after removing two electrons the configuration becomes: The electronic.
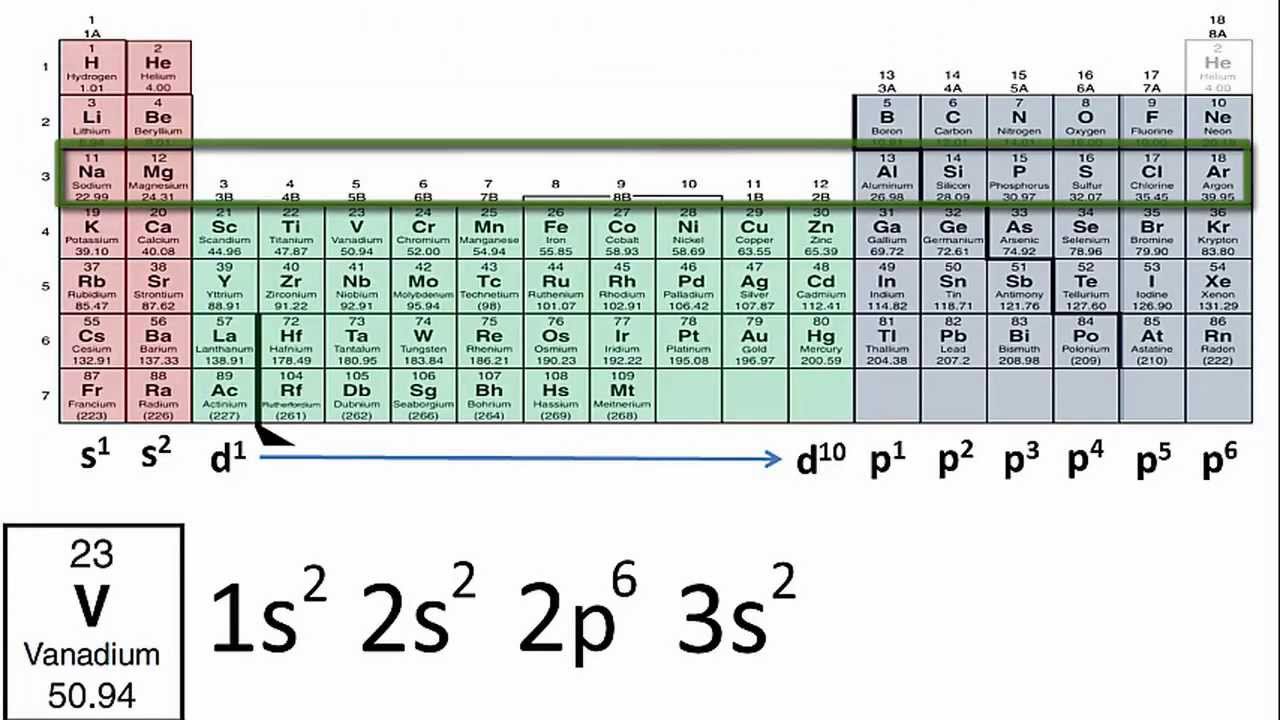
How to Write the Electron Configuration for Iron (Fe)?
The electronic configuration of fe is $\ce{[ar] 3d^6 4s^2}$. In order to write the iron electron configuration we first need to know the number of electrons for the fe atom (there are 26 electrons). The electron configuration of iron (fe) is [ar] 3d^6 4s^2. So after removing two electrons the configuration becomes: This means there are 26 electrons in an.
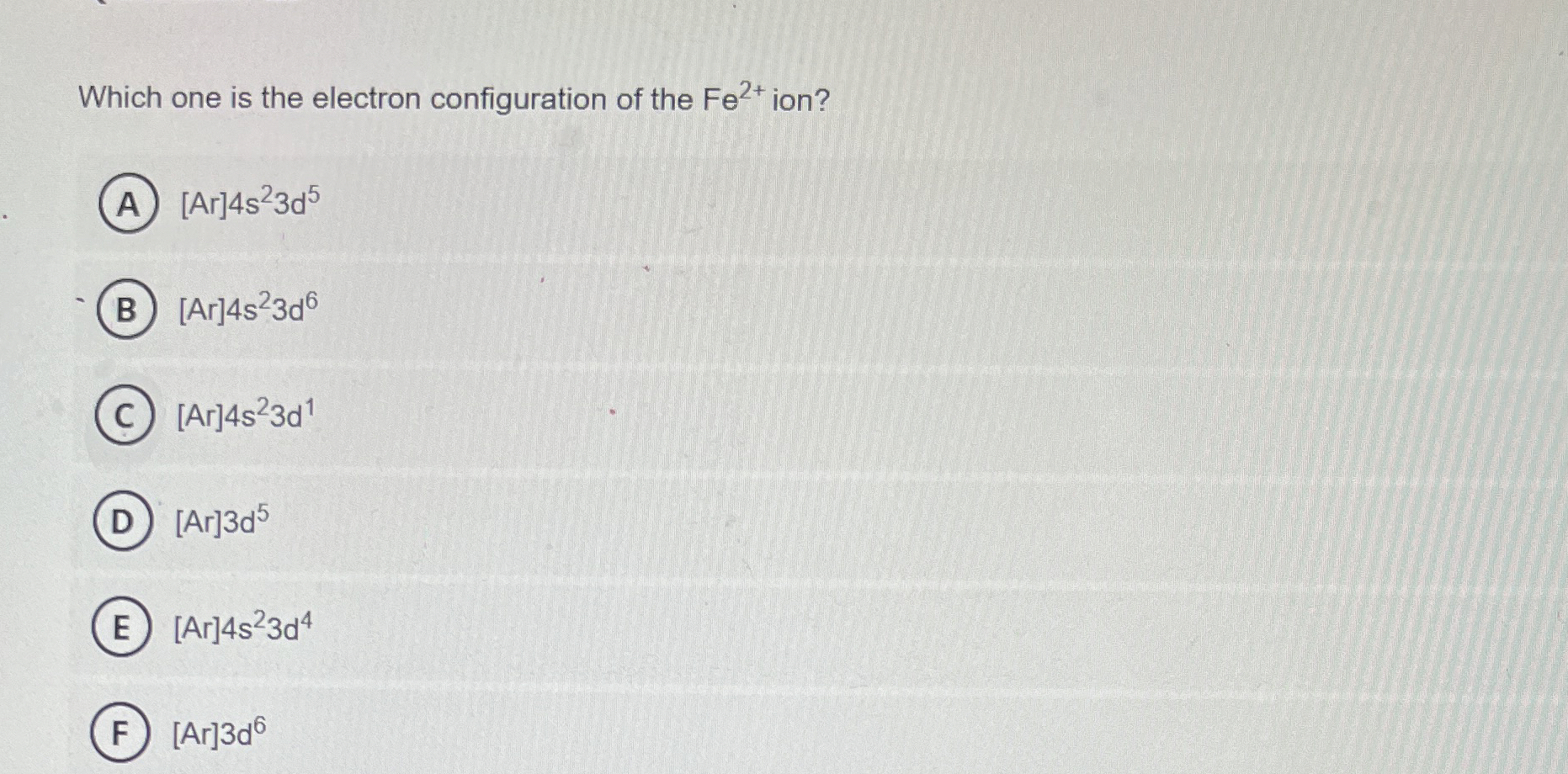
Solved Which one is the electron configuration of the Fe2+
So after removing two electrons the configuration becomes: This means there are 26 electrons in an iron atom, with 2 in the 4s. The electron configuration of iron (fe) is [ar] 3d^6 4s^2. The electronic configuration of fe is $\ce{[ar] 3d^6 4s^2}$. In order to write the iron electron configuration we first need to know the number of electrons for.
So After Removing Two Electrons The Configuration Becomes:
The electronic configuration of fe is $\ce{[ar] 3d^6 4s^2}$. The electron configuration of iron (fe) is [ar] 3d^6 4s^2. In order to write the iron electron configuration we first need to know the number of electrons for the fe atom (there are 26 electrons). This means there are 26 electrons in an iron atom, with 2 in the 4s.