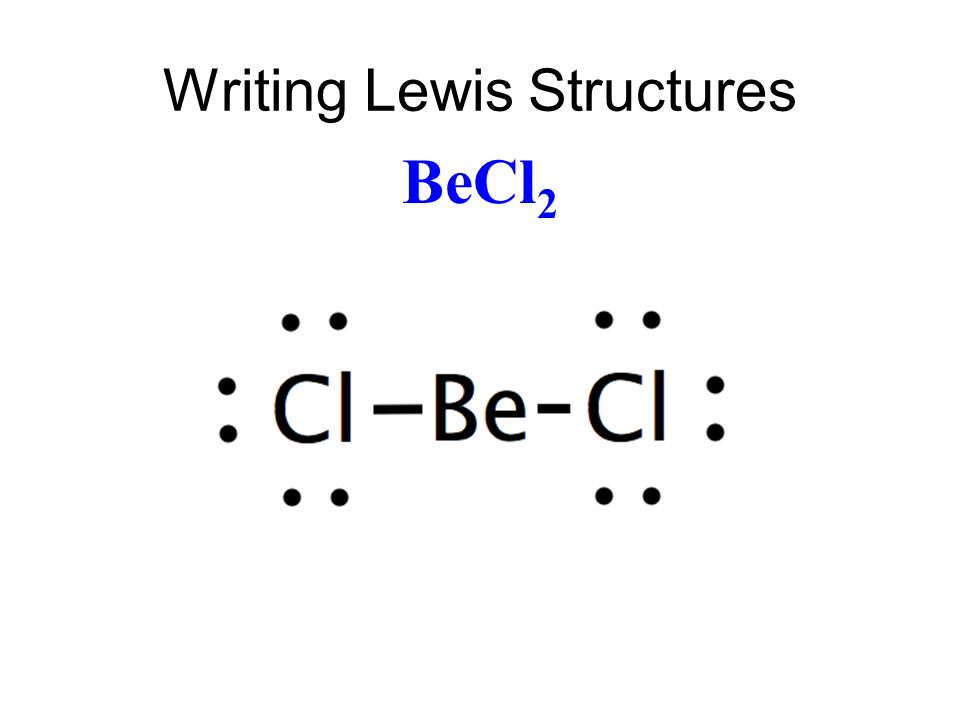
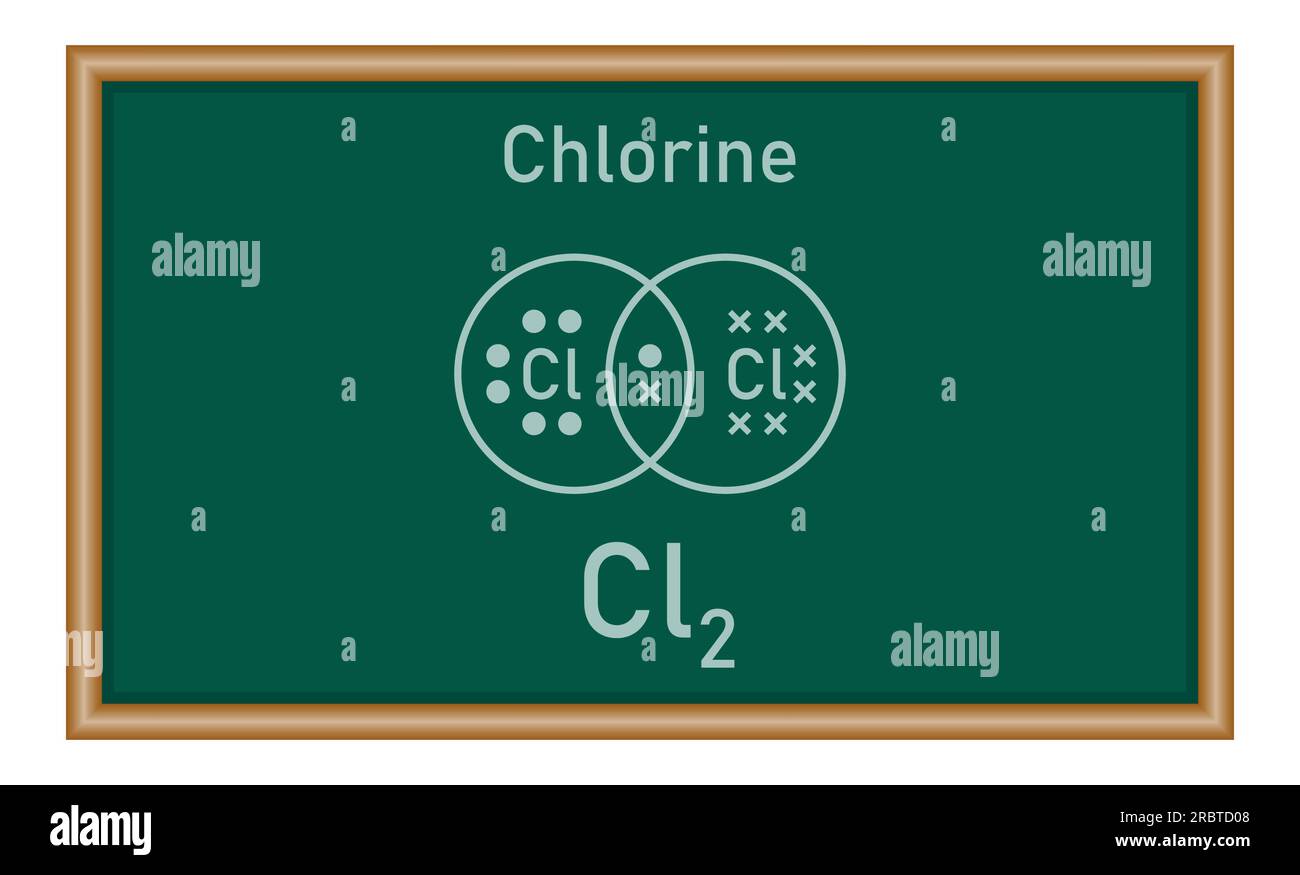
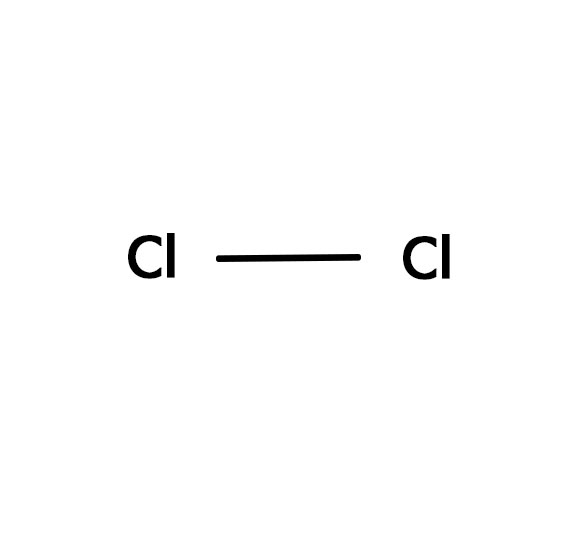What Is The Lewis Structure Of Cl2
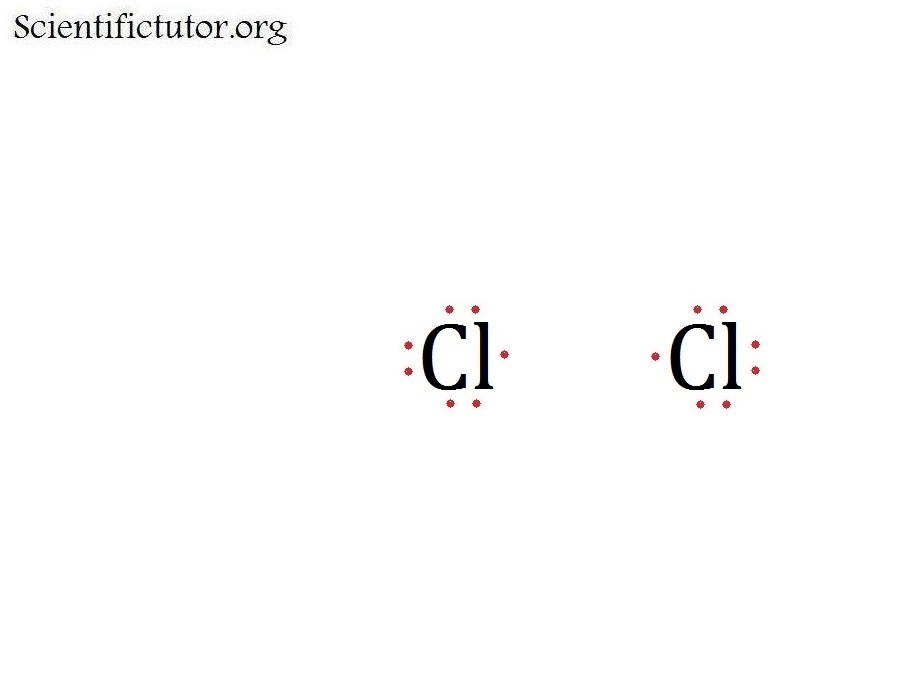
What Is The Lewis Structure Of Cl2 - Chlorine is a diatomic molecule and contains only two chlorine atoms. The lewis dot structure for cl2. Two things to keep in mind when drawing a lewis structure 1. All atoms must have 8 electrons 2.only use the valence electrons.
Two things to keep in mind when drawing a lewis structure 1. The lewis dot structure for cl2. All atoms must have 8 electrons 2.only use the valence electrons. Chlorine is a diatomic molecule and contains only two chlorine atoms.
The lewis dot structure for cl2. Two things to keep in mind when drawing a lewis structure 1. Chlorine is a diatomic molecule and contains only two chlorine atoms. All atoms must have 8 electrons 2.only use the valence electrons.
Cl2 Lewis Dot Structure
Two things to keep in mind when drawing a lewis structure 1. Chlorine is a diatomic molecule and contains only two chlorine atoms. All atoms must have 8 electrons 2.only use the valence electrons. The lewis dot structure for cl2.
Cl2 Lewis Structure, Geometry, Hybridization, and Polarity
Chlorine is a diatomic molecule and contains only two chlorine atoms. All atoms must have 8 electrons 2.only use the valence electrons. The lewis dot structure for cl2. Two things to keep in mind when drawing a lewis structure 1.
Clo2 1 Lewis Structure
The lewis dot structure for cl2. Chlorine is a diatomic molecule and contains only two chlorine atoms. Two things to keep in mind when drawing a lewis structure 1. All atoms must have 8 electrons 2.only use the valence electrons.
Lewis Structure For Cl2co
Chlorine is a diatomic molecule and contains only two chlorine atoms. The lewis dot structure for cl2. Two things to keep in mind when drawing a lewis structure 1. All atoms must have 8 electrons 2.only use the valence electrons.
Cl2 Lewis Dot Structure
The lewis dot structure for cl2. Two things to keep in mind when drawing a lewis structure 1. Chlorine is a diatomic molecule and contains only two chlorine atoms. All atoms must have 8 electrons 2.only use the valence electrons.
Cl Lewis Structure Diagram
The lewis dot structure for cl2. Chlorine is a diatomic molecule and contains only two chlorine atoms. All atoms must have 8 electrons 2.only use the valence electrons. Two things to keep in mind when drawing a lewis structure 1.
14+ Cl2 Lewis Structure Robhosking Diagram
Two things to keep in mind when drawing a lewis structure 1. All atoms must have 8 electrons 2.only use the valence electrons. The lewis dot structure for cl2. Chlorine is a diatomic molecule and contains only two chlorine atoms.
Clcn Lewis Structure
Two things to keep in mind when drawing a lewis structure 1. Chlorine is a diatomic molecule and contains only two chlorine atoms. All atoms must have 8 electrons 2.only use the valence electrons. The lewis dot structure for cl2.
C2cl2 Lewis Structure How To Draw The Lewis Structure For
Chlorine is a diatomic molecule and contains only two chlorine atoms. Two things to keep in mind when drawing a lewis structure 1. All atoms must have 8 electrons 2.only use the valence electrons. The lewis dot structure for cl2.
Two Things To Keep In Mind When Drawing A Lewis Structure 1.
All atoms must have 8 electrons 2.only use the valence electrons. The lewis dot structure for cl2. Chlorine is a diatomic molecule and contains only two chlorine atoms.