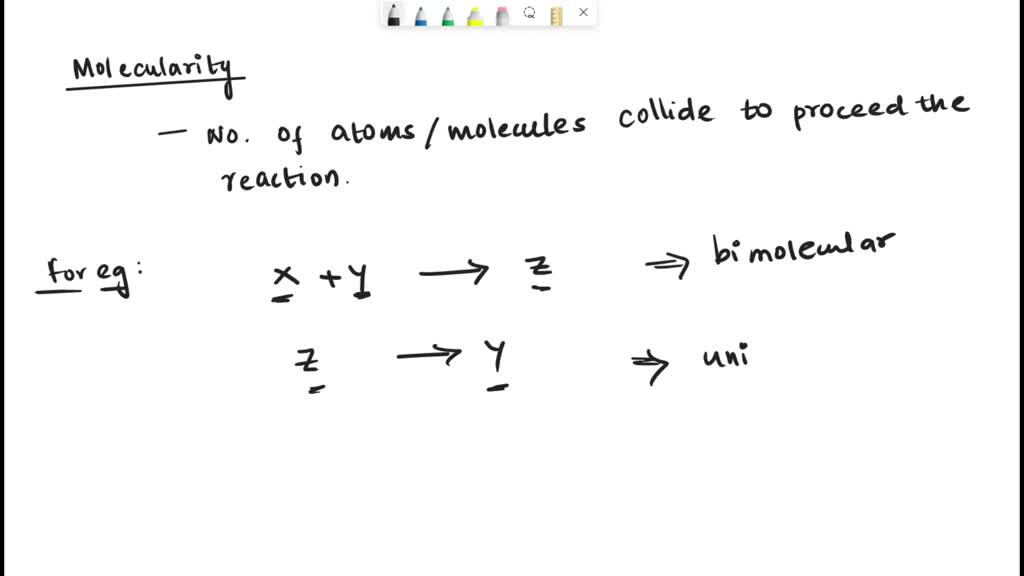What Is The Molecularity Of The Following Elementary Reaction
What Is The Molecularity Of The Following Elementary Reaction - An elementary reaction's rate law can directly be written from its molecularity. D) there is a greater proportion of reactants. It is equal to the sum. What is the molecularity of the following elementary reaction? The molecularity of a reaction is the number of molecules reacting in an elementary step. Molecularity of reaction (a) 2 no(g) + o2(g). A + 2b → ab₂. The molecularity of the elementary reaction 2no(g) n₂o₂(g) is 2 because there are two molecules of no involved in the reaction. Reactions can be unimolecular (one. Given the following elementary reaction what is the molecularity?
The molecularity of an elementary reaction is the number of molecules or atoms involved in the reaction. Reactions can be unimolecular (one. An elementary reaction's rate law can directly be written from its molecularity. A + 2b → ab₂. D) there is a greater proportion of reactants. The molecularity of the elementary reaction 2no(g) n₂o₂(g) is 2 because there are two molecules of no involved in the reaction. It is equal to the sum. What is the molecularity of the following elementary reaction? Molecularity is the number of reacting species involved in simultaneous collisions in an elementary or simplest reaction. Molecularity of reaction (a) 2 no(g) + o2(g).
A + 2b → ab₂. Molecularity of reaction (a) 2 no(g) + o2(g). Molecularity is the number of reacting species involved in simultaneous collisions in an elementary or simplest reaction. Given the following elementary reaction what is the molecularity? The molecularity of a reaction is the number of molecules reacting in an elementary step. An elementary reaction's rate law can directly be written from its molecularity. D) there is a greater proportion of reactants. The molecularity of an elementary reaction is the number of molecules or atoms involved in the reaction. The molecularity of the elementary reaction 2no(g) n₂o₂(g) is 2 because there are two molecules of no involved in the reaction. Reactions can be unimolecular (one.
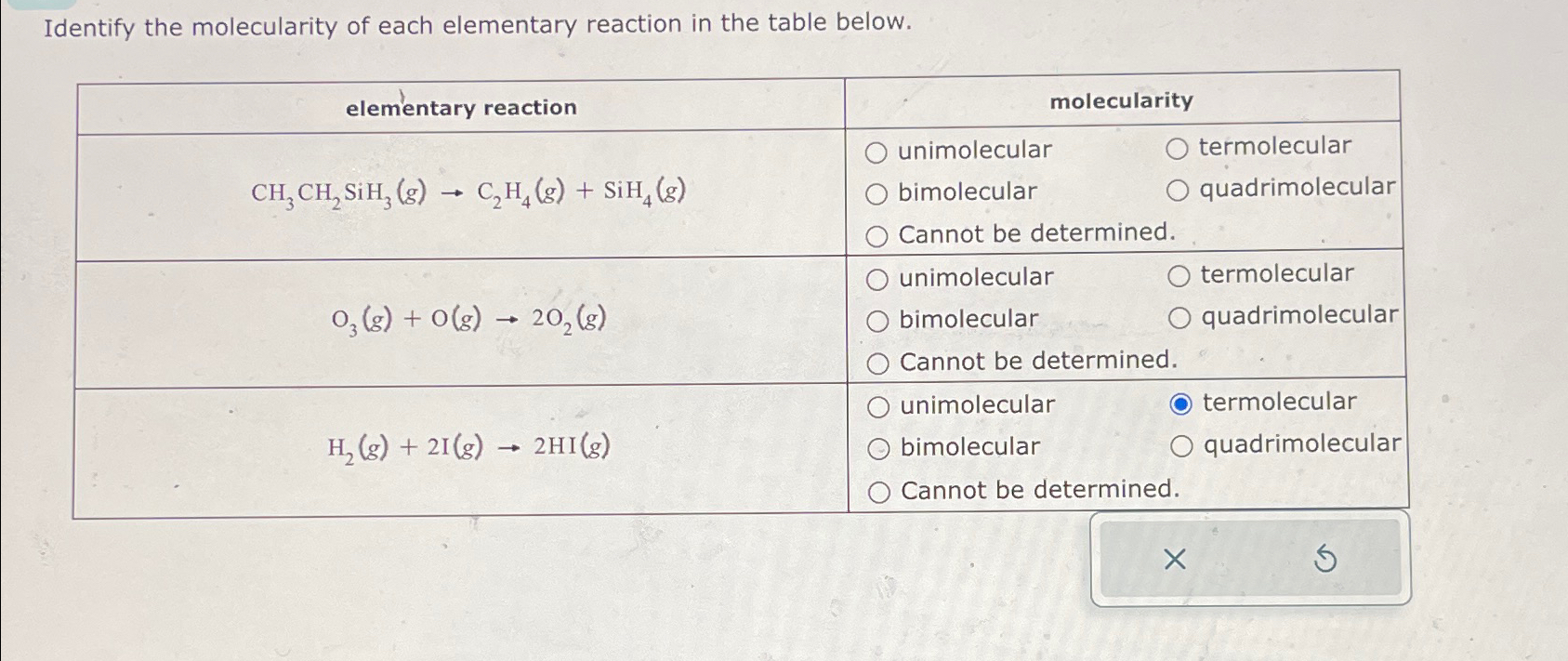
SOLVED Identify the molecularity of each elementary reaction in the
The molecularity of a reaction is the number of molecules reacting in an elementary step. A + 2b → ab₂. The molecularity of an elementary reaction is the number of molecules or atoms involved in the reaction. What is the molecularity of the following elementary reaction? Given the following elementary reaction what is the molecularity?
Solved Identify the molecularity of each elementary reaction
What is the molecularity of the following elementary reaction? The molecularity of a reaction is the number of molecules reacting in an elementary step. D) there is a greater proportion of reactants. An elementary reaction's rate law can directly be written from its molecularity. Reactions can be unimolecular (one.
Solved Identify the molecularity of each elementary reaction
The molecularity of a reaction is the number of molecules reacting in an elementary step. D) there is a greater proportion of reactants. Molecularity of reaction (a) 2 no(g) + o2(g). It is equal to the sum. The molecularity of the elementary reaction 2no(g) n₂o₂(g) is 2 because there are two molecules of no involved in the reaction.
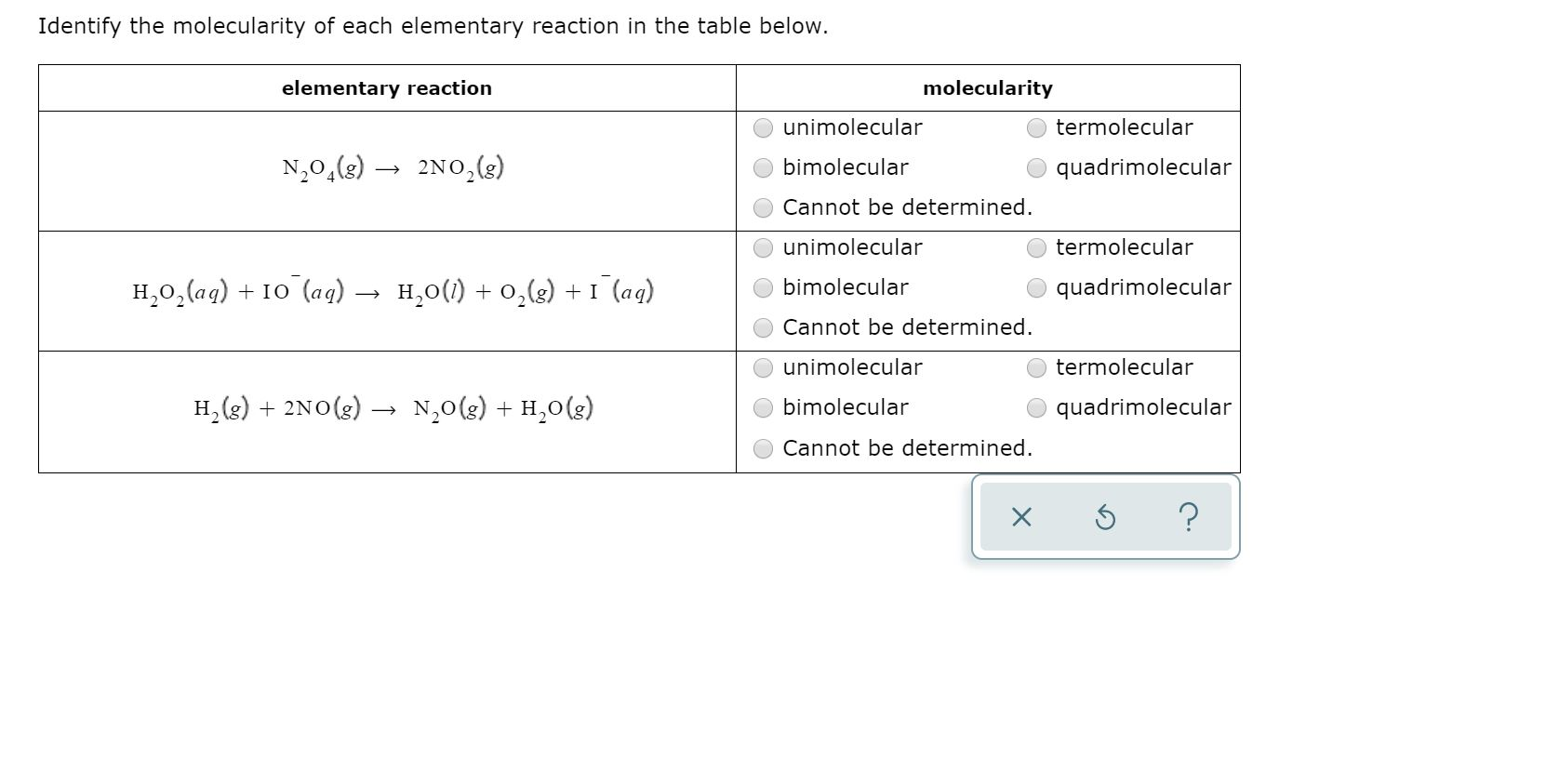
Solved Identify the molecularity of each elementary reaction
The molecularity of a reaction is the number of molecules reacting in an elementary step. Molecularity of reaction (a) 2 no(g) + o2(g). A + 2b → ab₂. An elementary reaction's rate law can directly be written from its molecularity. D) there is a greater proportion of reactants.
Solved Identify the molecularity of each elementary reaction
The molecularity of a reaction is the number of molecules reacting in an elementary step. Molecularity of reaction (a) 2 no(g) + o2(g). D) there is a greater proportion of reactants. The molecularity of an elementary reaction is the number of molecules or atoms involved in the reaction. Molecularity is the number of reacting species involved in simultaneous collisions in.
Elementary Reaction Definition and Examples (Chemistry)
An elementary reaction's rate law can directly be written from its molecularity. Given the following elementary reaction what is the molecularity? D) there is a greater proportion of reactants. The molecularity of a reaction is the number of molecules reacting in an elementary step. The molecularity of an elementary reaction is the number of molecules or atoms involved in the.
Solved Identify the molecularity of each elementary reaction
Given the following elementary reaction what is the molecularity? What is the molecularity of the following elementary reaction? Molecularity is the number of reacting species involved in simultaneous collisions in an elementary or simplest reaction. The molecularity of a reaction is the number of molecules reacting in an elementary step. An elementary reaction's rate law can directly be written from.
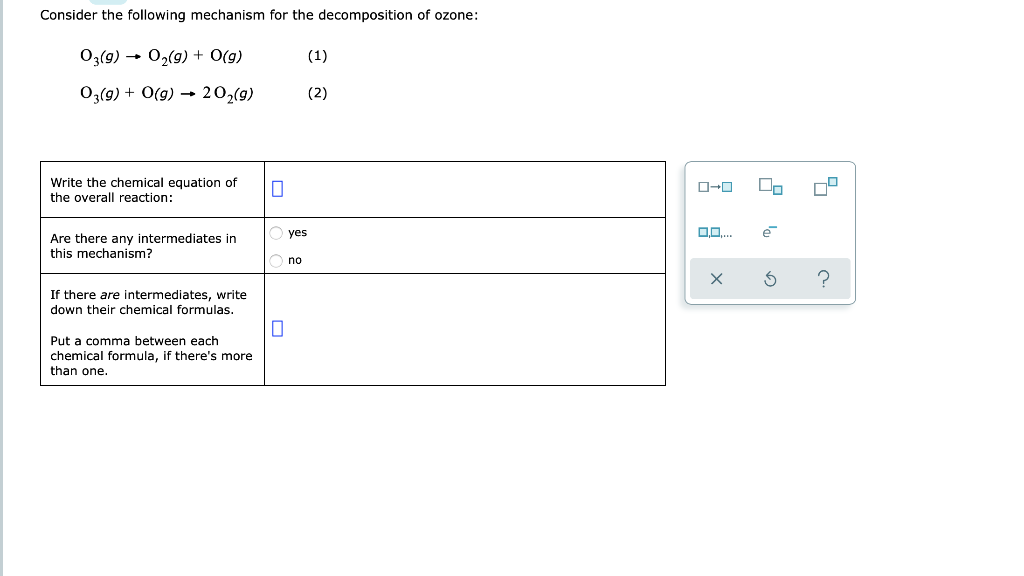
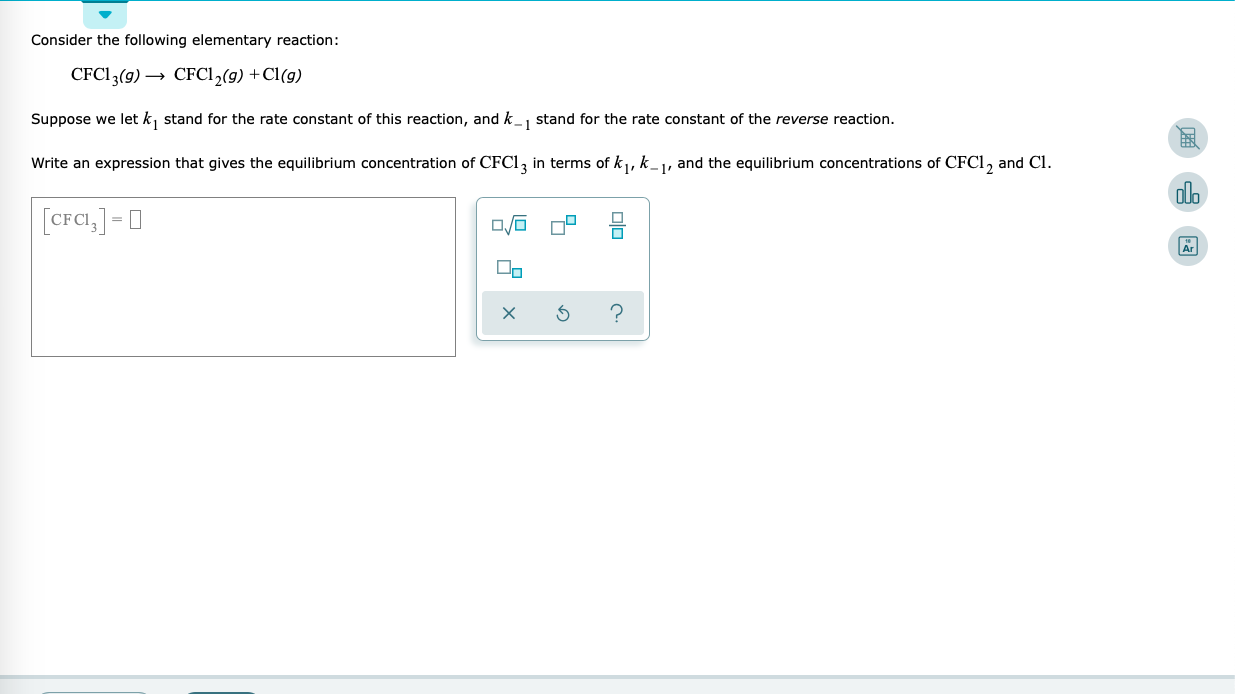
Solved Consider the following
What is the molecularity of the following elementary reaction? Given the following elementary reaction what is the molecularity? D) there is a greater proportion of reactants. The molecularity of the elementary reaction 2no(g) n₂o₂(g) is 2 because there are two molecules of no involved in the reaction. The molecularity of an elementary reaction is the number of molecules or atoms.
Solved Identify The Molecularity Of Each Elementary React...
D) there is a greater proportion of reactants. Reactions can be unimolecular (one. The molecularity of an elementary reaction is the number of molecules or atoms involved in the reaction. The molecularity of a reaction is the number of molecules reacting in an elementary step. It is equal to the sum.
Solved Identify the molecularity of each elementary reaction
Molecularity of reaction (a) 2 no(g) + o2(g). Molecularity is the number of reacting species involved in simultaneous collisions in an elementary or simplest reaction. The molecularity of an elementary reaction is the number of molecules or atoms involved in the reaction. The molecularity of the elementary reaction 2no(g) n₂o₂(g) is 2 because there are two molecules of no involved.
It Is Equal To The Sum.
Given the following elementary reaction what is the molecularity? A + 2b → ab₂. Molecularity is the number of reacting species involved in simultaneous collisions in an elementary or simplest reaction. An elementary reaction's rate law can directly be written from its molecularity.
The Molecularity Of The Elementary Reaction 2No(G) N₂O₂(G) Is 2 Because There Are Two Molecules Of No Involved In The Reaction.
D) there is a greater proportion of reactants. Reactions can be unimolecular (one. Molecularity of reaction (a) 2 no(g) + o2(g). What is the molecularity of the following elementary reaction?
The Molecularity Of A Reaction Is The Number Of Molecules Reacting In An Elementary Step.
The molecularity of an elementary reaction is the number of molecules or atoms involved in the reaction.