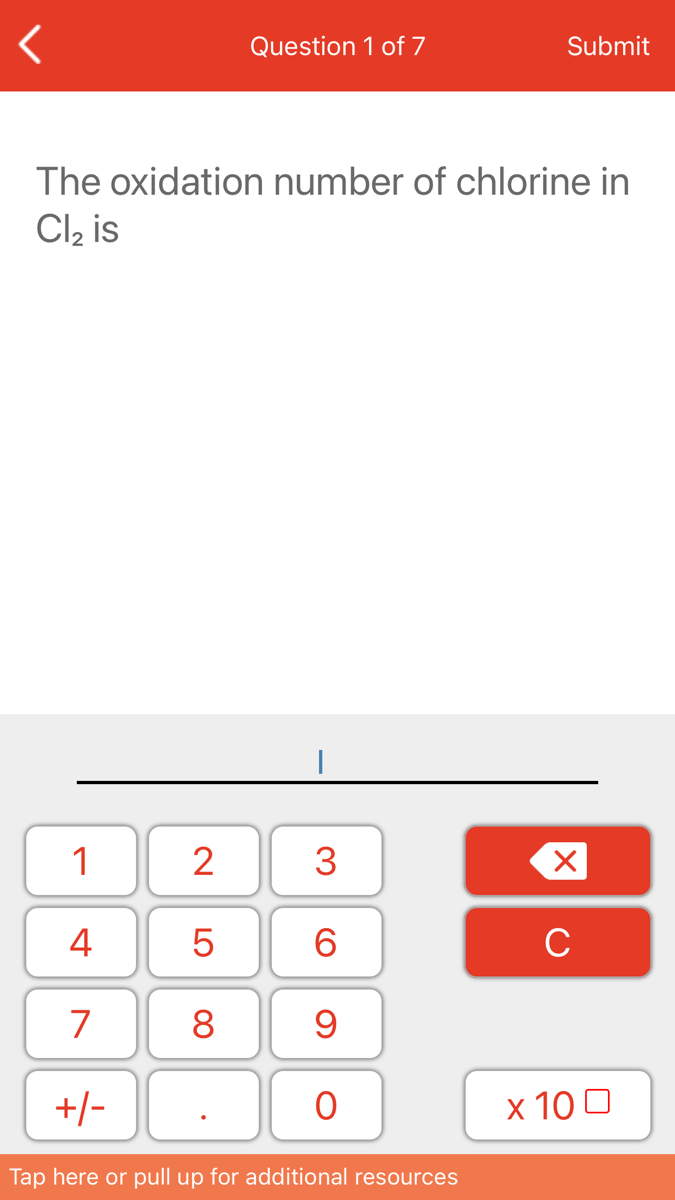
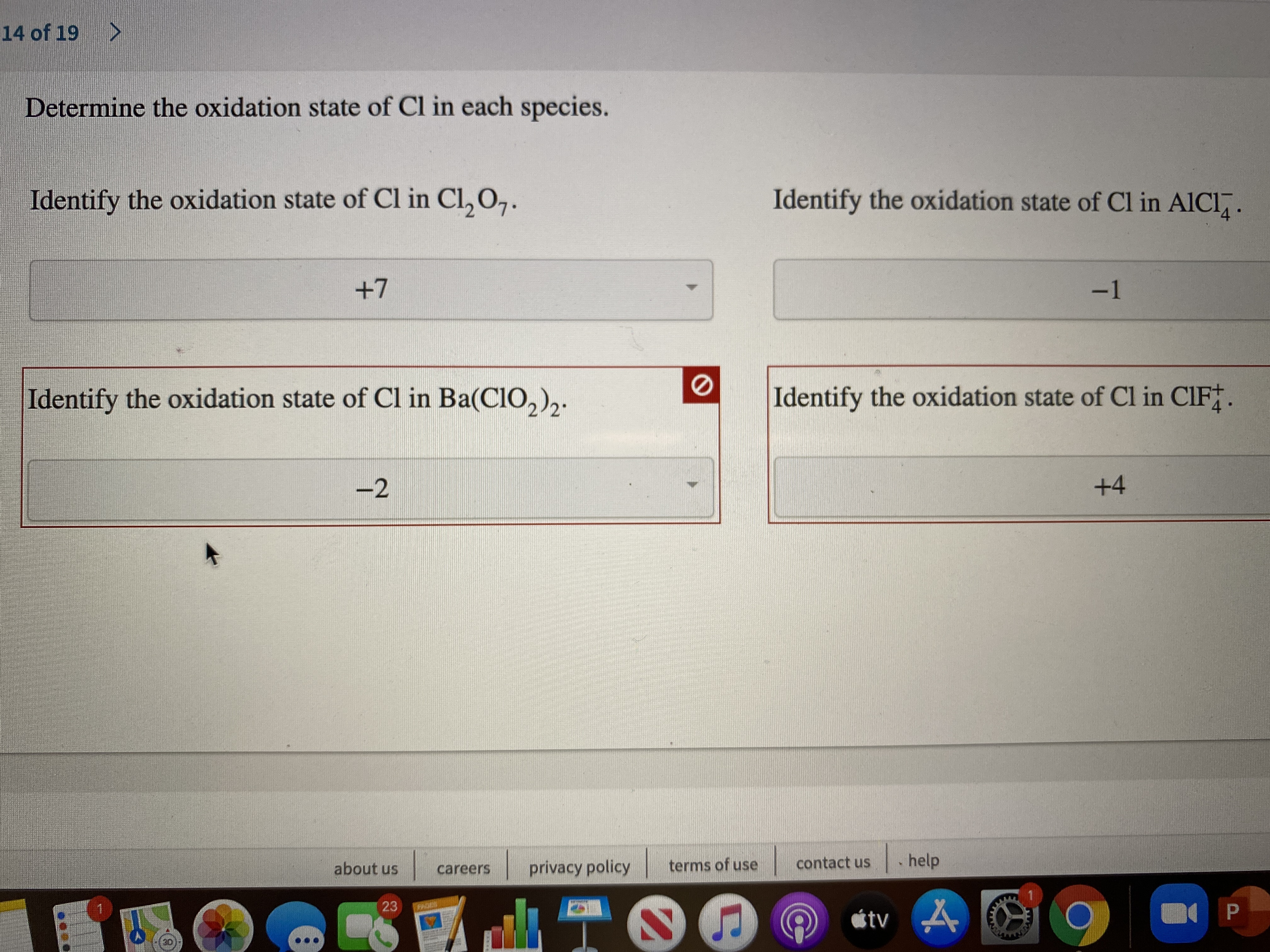
What Is The Oxidation Number Of Cl In Clo4
What Is The Oxidation Number Of Cl In Clo4 - The oxidation state of chlorine (cl) in the perchlorate ion (clo₄⁻) is +7. This is determined by considering the overall charge of. The oxidation number indicates the degree of oxidation (loss of electrons) of an atom in a chemical compound. The oxidation number of chlorine in clo₄ is +7. More information the oxidation state of +7 for chlorine is its highest oxidation state, indicating.
The oxidation number indicates the degree of oxidation (loss of electrons) of an atom in a chemical compound. The oxidation state of chlorine (cl) in the perchlorate ion (clo₄⁻) is +7. This is determined by considering the overall charge of. The oxidation number of chlorine in clo₄ is +7. More information the oxidation state of +7 for chlorine is its highest oxidation state, indicating.
This is determined by considering the overall charge of. The oxidation state of chlorine (cl) in the perchlorate ion (clo₄⁻) is +7. The oxidation number indicates the degree of oxidation (loss of electrons) of an atom in a chemical compound. More information the oxidation state of +7 for chlorine is its highest oxidation state, indicating. The oxidation number of chlorine in clo₄ is +7.
Oxidation Number Rules Chart
The oxidation state of chlorine (cl) in the perchlorate ion (clo₄⁻) is +7. The oxidation number of chlorine in clo₄ is +7. The oxidation number indicates the degree of oxidation (loss of electrons) of an atom in a chemical compound. More information the oxidation state of +7 for chlorine is its highest oxidation state, indicating. This is determined by considering.
Solved B. Determine the oxidation number of Cl a) HClO4 b)
The oxidation number indicates the degree of oxidation (loss of electrons) of an atom in a chemical compound. More information the oxidation state of +7 for chlorine is its highest oxidation state, indicating. This is determined by considering the overall charge of. The oxidation state of chlorine (cl) in the perchlorate ion (clo₄⁻) is +7. The oxidation number of chlorine.
Answered The oxidation number of chlorine in Cl2… bartleby
This is determined by considering the overall charge of. The oxidation state of chlorine (cl) in the perchlorate ion (clo₄⁻) is +7. The oxidation number of chlorine in clo₄ is +7. The oxidation number indicates the degree of oxidation (loss of electrons) of an atom in a chemical compound. More information the oxidation state of +7 for chlorine is its.
Formation of ClO3⁻ and ClO4⁻ during the electrochemical oxidation of
This is determined by considering the overall charge of. The oxidation state of chlorine (cl) in the perchlorate ion (clo₄⁻) is +7. More information the oxidation state of +7 for chlorine is its highest oxidation state, indicating. The oxidation number of chlorine in clo₄ is +7. The oxidation number indicates the degree of oxidation (loss of electrons) of an atom.
Oxidation Number of Chlorine LinatuSims
The oxidation number indicates the degree of oxidation (loss of electrons) of an atom in a chemical compound. The oxidation number of chlorine in clo₄ is +7. This is determined by considering the overall charge of. More information the oxidation state of +7 for chlorine is its highest oxidation state, indicating. The oxidation state of chlorine (cl) in the perchlorate.
Solved Determine the oxidation number of Cl in each of the
More information the oxidation state of +7 for chlorine is its highest oxidation state, indicating. The oxidation number indicates the degree of oxidation (loss of electrons) of an atom in a chemical compound. The oxidation number of chlorine in clo₄ is +7. The oxidation state of chlorine (cl) in the perchlorate ion (clo₄⁻) is +7. This is determined by considering.
[Solved] Determine the oxidation state of Cl Cl in each species
This is determined by considering the overall charge of. More information the oxidation state of +7 for chlorine is its highest oxidation state, indicating. The oxidation state of chlorine (cl) in the perchlorate ion (clo₄⁻) is +7. The oxidation number of chlorine in clo₄ is +7. The oxidation number indicates the degree of oxidation (loss of electrons) of an atom.
[Solved] Determine the oxidation state of Cl Cl in each species
The oxidation state of chlorine (cl) in the perchlorate ion (clo₄⁻) is +7. The oxidation number indicates the degree of oxidation (loss of electrons) of an atom in a chemical compound. More information the oxidation state of +7 for chlorine is its highest oxidation state, indicating. The oxidation number of chlorine in clo₄ is +7. This is determined by considering.
What is the oxidation number of Cl in CaOCl2 Explain with structure
The oxidation number indicates the degree of oxidation (loss of electrons) of an atom in a chemical compound. The oxidation number of chlorine in clo₄ is +7. The oxidation state of chlorine (cl) in the perchlorate ion (clo₄⁻) is +7. More information the oxidation state of +7 for chlorine is its highest oxidation state, indicating. This is determined by considering.
Answered Determine the oxidation state of Cl in… bartleby
The oxidation number indicates the degree of oxidation (loss of electrons) of an atom in a chemical compound. The oxidation state of chlorine (cl) in the perchlorate ion (clo₄⁻) is +7. This is determined by considering the overall charge of. The oxidation number of chlorine in clo₄ is +7. More information the oxidation state of +7 for chlorine is its.
This Is Determined By Considering The Overall Charge Of.
The oxidation number indicates the degree of oxidation (loss of electrons) of an atom in a chemical compound. The oxidation number of chlorine in clo₄ is +7. The oxidation state of chlorine (cl) in the perchlorate ion (clo₄⁻) is +7. More information the oxidation state of +7 for chlorine is its highest oxidation state, indicating.