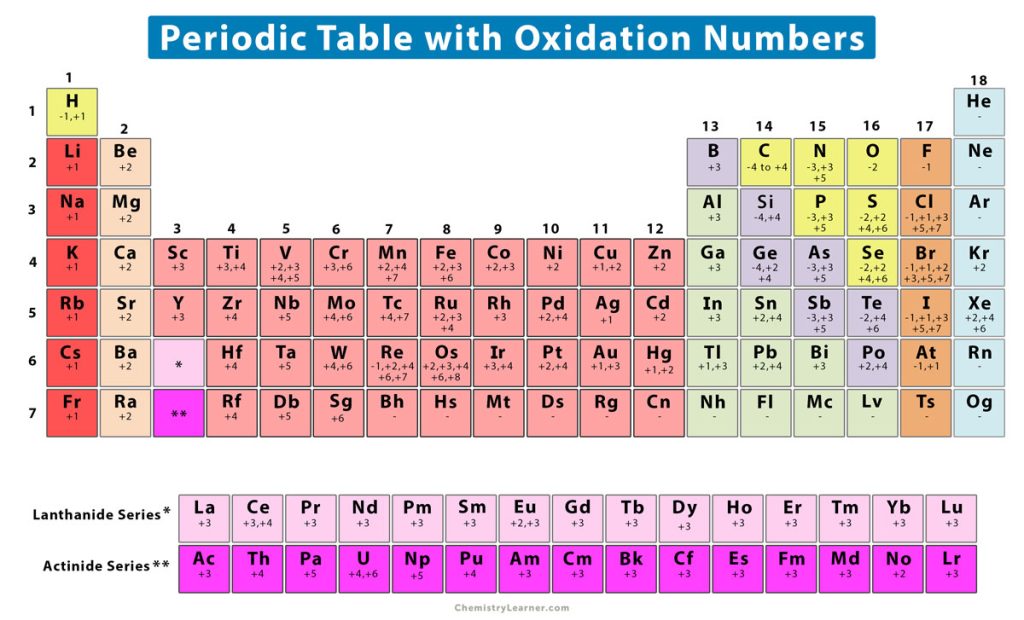
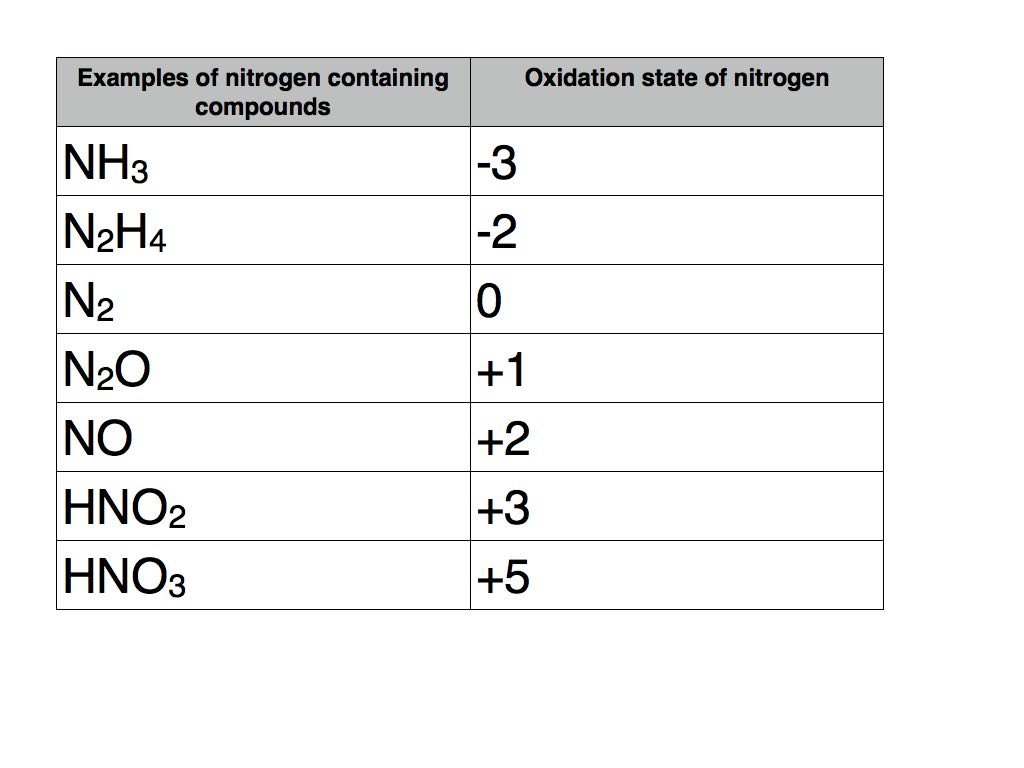
What Is The Oxidation State Of N In Nh4
What Is The Oxidation State Of N In Nh4 - What is the oxidation state of hydrogen in (nh4)+? The average oxidation state of the 4 h atoms in (nh 4) + is +1. Enter the formula of a. This is because the total positive charge contributed by the four.
This is because the total positive charge contributed by the four. What is the oxidation state of hydrogen in (nh4)+? The average oxidation state of the 4 h atoms in (nh 4) + is +1. Enter the formula of a.
The average oxidation state of the 4 h atoms in (nh 4) + is +1. Enter the formula of a. This is because the total positive charge contributed by the four. What is the oxidation state of hydrogen in (nh4)+?
Oxidation state nitrogen is incorrectly given compound oxidation state
What is the oxidation state of hydrogen in (nh4)+? Enter the formula of a. This is because the total positive charge contributed by the four. The average oxidation state of the 4 h atoms in (nh 4) + is +1.
Oxidation Number (State) Definition, Rules, How to Find, and Examples
This is because the total positive charge contributed by the four. The average oxidation state of the 4 h atoms in (nh 4) + is +1. Enter the formula of a. What is the oxidation state of hydrogen in (nh4)+?
Oxidation state examples Online Chemistry Tutor
Enter the formula of a. What is the oxidation state of hydrogen in (nh4)+? The average oxidation state of the 4 h atoms in (nh 4) + is +1. This is because the total positive charge contributed by the four.
Oxidation state of nitrogen is incorrectly given in
What is the oxidation state of hydrogen in (nh4)+? The average oxidation state of the 4 h atoms in (nh 4) + is +1. Enter the formula of a. This is because the total positive charge contributed by the four.
Oxidation state nitrogen is incorrectly given compound oxidation state
What is the oxidation state of hydrogen in (nh4)+? Enter the formula of a. This is because the total positive charge contributed by the four. The average oxidation state of the 4 h atoms in (nh 4) + is +1.
Oxidation State Of N
This is because the total positive charge contributed by the four. Enter the formula of a. The average oxidation state of the 4 h atoms in (nh 4) + is +1. What is the oxidation state of hydrogen in (nh4)+?
Oxidation State Of N
What is the oxidation state of hydrogen in (nh4)+? Enter the formula of a. The average oxidation state of the 4 h atoms in (nh 4) + is +1. This is because the total positive charge contributed by the four.
Oxidation Rules Chart
What is the oxidation state of hydrogen in (nh4)+? This is because the total positive charge contributed by the four. The average oxidation state of the 4 h atoms in (nh 4) + is +1. Enter the formula of a.
Oxidation state nitrogen is incorrectly given compound oxidation state
Enter the formula of a. The average oxidation state of the 4 h atoms in (nh 4) + is +1. What is the oxidation state of hydrogen in (nh4)+? This is because the total positive charge contributed by the four.
What Is The Oxidation State Of Hydrogen In (Nh4)+?
The average oxidation state of the 4 h atoms in (nh 4) + is +1. Enter the formula of a. This is because the total positive charge contributed by the four.