What Is The Ph Of A 0 04 M Koh Solution
What Is The Ph Of A 0 04 M Koh Solution - Determining the ph of a 0.04 m koh solution to. Koh is a strong base, which means that it will have a high ph in aqueous solution. The ph of a 0.04 m koh solution is 12. Stuck on a stem question? Here’s the best way to solve it. Post your question and get video answers from professional experts: Use the concentration of k o h, which is 0.04 m, to find the concentration of hydroxide ions, [o h −].
Determining the ph of a 0.04 m koh solution to. Koh is a strong base, which means that it will have a high ph in aqueous solution. Post your question and get video answers from professional experts: Here’s the best way to solve it. Use the concentration of k o h, which is 0.04 m, to find the concentration of hydroxide ions, [o h −]. Stuck on a stem question? The ph of a 0.04 m koh solution is 12.
Determining the ph of a 0.04 m koh solution to. The ph of a 0.04 m koh solution is 12. Stuck on a stem question? Post your question and get video answers from professional experts: Koh is a strong base, which means that it will have a high ph in aqueous solution. Use the concentration of k o h, which is 0.04 m, to find the concentration of hydroxide ions, [o h −]. Here’s the best way to solve it.
Solved T(°C) Calculate the pH of a 0.0201 M KOH solution at
Determining the ph of a 0.04 m koh solution to. Use the concentration of k o h, which is 0.04 m, to find the concentration of hydroxide ions, [o h −]. Koh is a strong base, which means that it will have a high ph in aqueous solution. Stuck on a stem question? Here’s the best way to solve it.
Solved 5. Calculate the pH of the 2M KOH solution?
Stuck on a stem question? Koh is a strong base, which means that it will have a high ph in aqueous solution. Use the concentration of k o h, which is 0.04 m, to find the concentration of hydroxide ions, [o h −]. Determining the ph of a 0.04 m koh solution to. Post your question and get video answers.
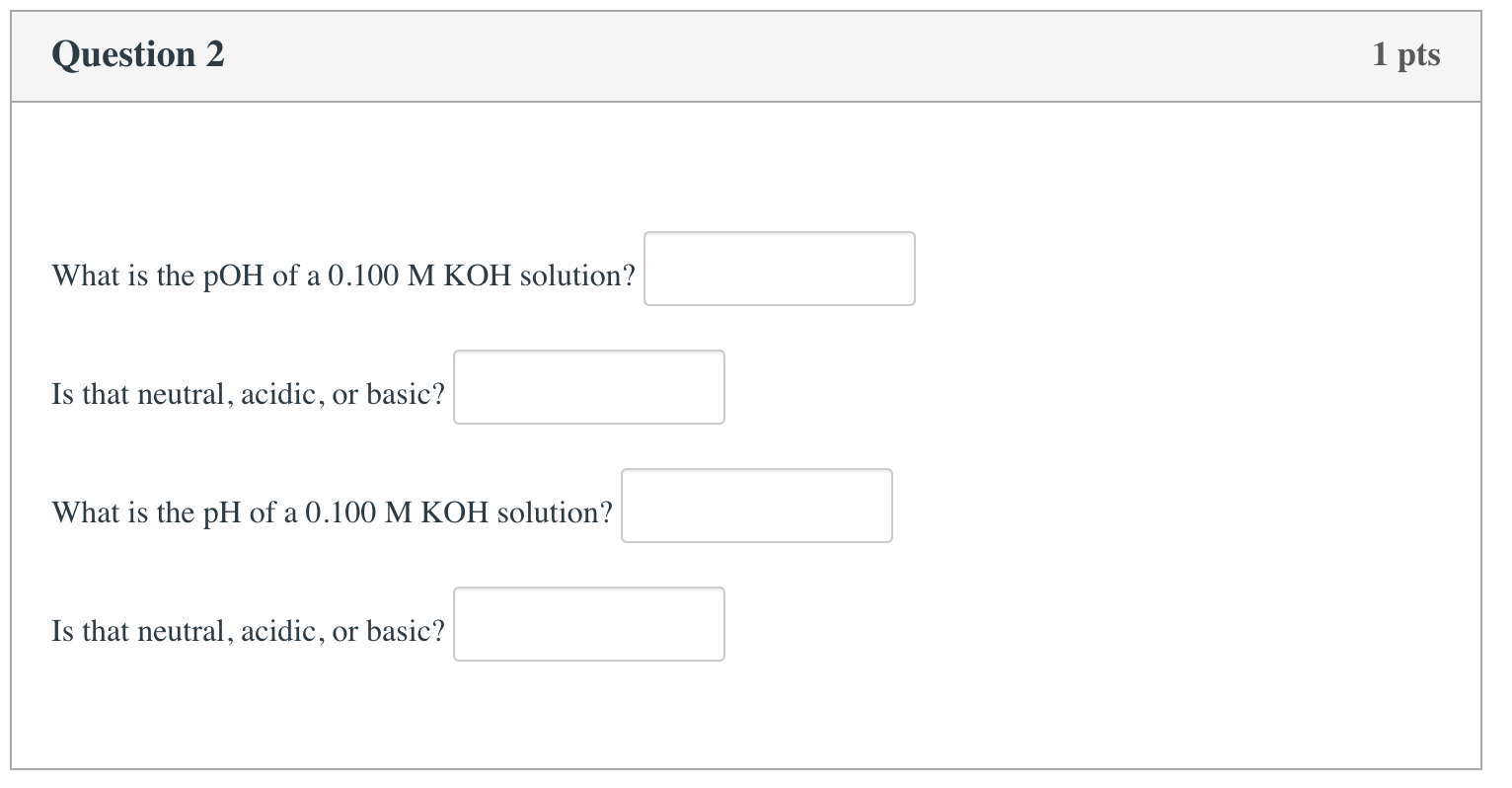
Solved Question 2 1 pts What is the pOH of a 0.100 M KOH
Determining the ph of a 0.04 m koh solution to. Post your question and get video answers from professional experts: Koh is a strong base, which means that it will have a high ph in aqueous solution. Here’s the best way to solve it. Use the concentration of k o h, which is 0.04 m, to find the concentration of.
Solved Calculate the pH of 0.01 M KOH solution. (KOH is
Use the concentration of k o h, which is 0.04 m, to find the concentration of hydroxide ions, [o h −]. Koh is a strong base, which means that it will have a high ph in aqueous solution. Determining the ph of a 0.04 m koh solution to. The ph of a 0.04 m koh solution is 12. Stuck on.
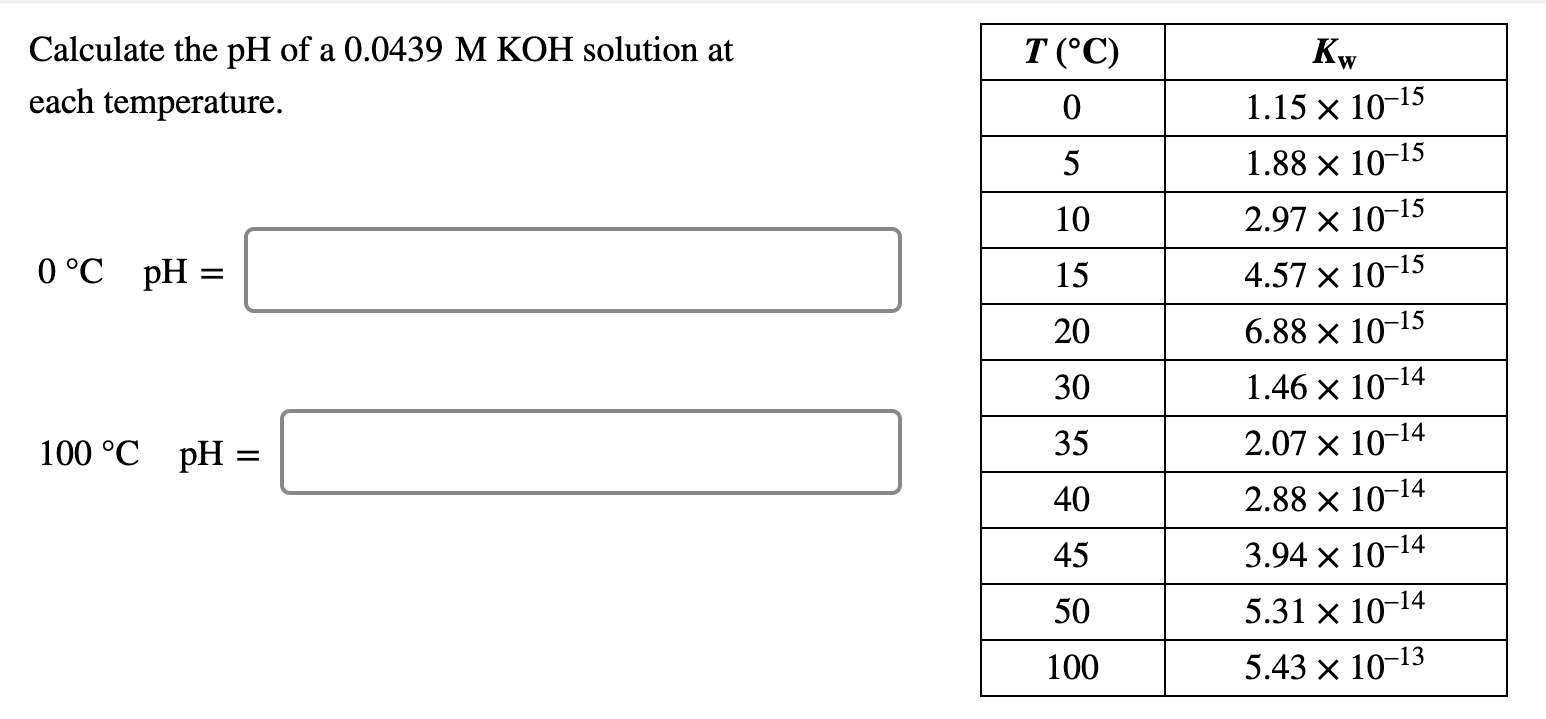
Solved Calculate the pH of a 0.0439 M KOH solution at each
Stuck on a stem question? Use the concentration of k o h, which is 0.04 m, to find the concentration of hydroxide ions, [o h −]. Determining the ph of a 0.04 m koh solution to. Here’s the best way to solve it. Post your question and get video answers from professional experts:
SOLUTION Calculate the pH of a 0 58 M KOH solutionSolutionpOH = log(0
Post your question and get video answers from professional experts: Stuck on a stem question? Determining the ph of a 0.04 m koh solution to. The ph of a 0.04 m koh solution is 12. Here’s the best way to solve it.
Solved T("C) Calculate the pH of a 0.0464 M KOH solution at
Here’s the best way to solve it. Koh is a strong base, which means that it will have a high ph in aqueous solution. Post your question and get video answers from professional experts: Use the concentration of k o h, which is 0.04 m, to find the concentration of hydroxide ions, [o h −]. Determining the ph of a.
Solved A 50.0 mL solution of 0.165 M KOH is titrated with
Here’s the best way to solve it. The ph of a 0.04 m koh solution is 12. Use the concentration of k o h, which is 0.04 m, to find the concentration of hydroxide ions, [o h −]. Stuck on a stem question? Post your question and get video answers from professional experts:
At 90 ^o C, the pH of 0.001M KOH solution will be
Post your question and get video answers from professional experts: The ph of a 0.04 m koh solution is 12. Here’s the best way to solve it. Koh is a strong base, which means that it will have a high ph in aqueous solution. Use the concentration of k o h, which is 0.04 m, to find the concentration of.
Use The Concentration Of K O H, Which Is 0.04 M, To Find The Concentration Of Hydroxide Ions, [O H −].
Determining the ph of a 0.04 m koh solution to. The ph of a 0.04 m koh solution is 12. Post your question and get video answers from professional experts: Stuck on a stem question?
Koh Is A Strong Base, Which Means That It Will Have A High Ph In Aqueous Solution.
Here’s the best way to solve it.