What Is True When A Reaction Has Reached Equilibrium
What Is True When A Reaction Has Reached Equilibrium - When a chemical reaction has reached equilibrium are products and reactants being formed? What is true when a reaction had reached equilibrium? When a reaction has reached equilibrium, the correct statement is a: The reaction rate is equal in both directions. When a reaction has reached equilibrium, it means that the rates of the forward and reverse reactions are equal. Yes, the forward reaction and reverse reaction occur but. In a chemical reaction, a proper equilibrium is generally reached when the total rate of the forward reaction equals the rate of. This does not mean that the reaction.
This does not mean that the reaction. When a reaction has reached equilibrium, the correct statement is a: The reaction rate is equal in both directions. When a reaction has reached equilibrium, it means that the rates of the forward and reverse reactions are equal. What is true when a reaction had reached equilibrium? Yes, the forward reaction and reverse reaction occur but. When a chemical reaction has reached equilibrium are products and reactants being formed? In a chemical reaction, a proper equilibrium is generally reached when the total rate of the forward reaction equals the rate of.
When a chemical reaction has reached equilibrium are products and reactants being formed? In a chemical reaction, a proper equilibrium is generally reached when the total rate of the forward reaction equals the rate of. The reaction rate is equal in both directions. When a reaction has reached equilibrium, the correct statement is a: What is true when a reaction had reached equilibrium? This does not mean that the reaction. Yes, the forward reaction and reverse reaction occur but. When a reaction has reached equilibrium, it means that the rates of the forward and reverse reactions are equal.
(PPT) Section 8.3—Reaction Quotient How can we tell if a reaction has
When a reaction has reached equilibrium, the correct statement is a: The reaction rate is equal in both directions. This does not mean that the reaction. What is true when a reaction had reached equilibrium? In a chemical reaction, a proper equilibrium is generally reached when the total rate of the forward reaction equals the rate of.
Solved 3. The following reaction has reached equilibrium at
The reaction rate is equal in both directions. When a reaction has reached equilibrium, it means that the rates of the forward and reverse reactions are equal. In a chemical reaction, a proper equilibrium is generally reached when the total rate of the forward reaction equals the rate of. What is true when a reaction had reached equilibrium? This does.
Solved A reversible reaction has reached equilibrium when
When a chemical reaction has reached equilibrium are products and reactants being formed? In a chemical reaction, a proper equilibrium is generally reached when the total rate of the forward reaction equals the rate of. This does not mean that the reaction. What is true when a reaction had reached equilibrium? When a reaction has reached equilibrium, the correct statement.
What is true for the equilibrium reaction ? Filo
When a reaction has reached equilibrium, the correct statement is a: When a reaction has reached equilibrium, it means that the rates of the forward and reverse reactions are equal. This does not mean that the reaction. When a chemical reaction has reached equilibrium are products and reactants being formed? What is true when a reaction had reached equilibrium?
Solved The reaction reached equilibrium at a certain high
This does not mean that the reaction. When a reaction has reached equilibrium, the correct statement is a: In a chemical reaction, a proper equilibrium is generally reached when the total rate of the forward reaction equals the rate of. When a reaction has reached equilibrium, it means that the rates of the forward and reverse reactions are equal. The.
Solved QUESTION 1 Which of the following is true of a
When a reaction has reached equilibrium, the correct statement is a: The reaction rate is equal in both directions. What is true when a reaction had reached equilibrium? When a reaction has reached equilibrium, it means that the rates of the forward and reverse reactions are equal. In a chemical reaction, a proper equilibrium is generally reached when the total.
SOLVED Chemical equilibrium is reached when . hints chemical
When a reaction has reached equilibrium, it means that the rates of the forward and reverse reactions are equal. What is true when a reaction had reached equilibrium? When a chemical reaction has reached equilibrium are products and reactants being formed? The reaction rate is equal in both directions. This does not mean that the reaction.
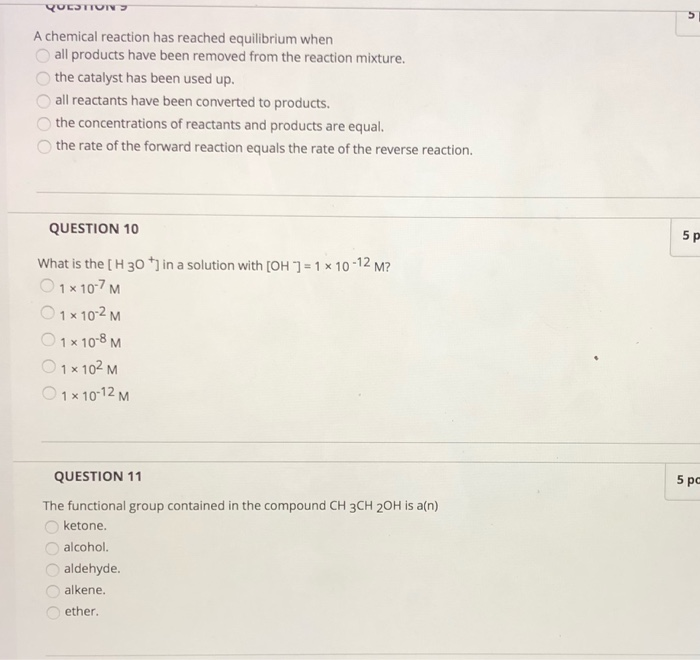
Solved QUESTION A chemical reaction has reached equilibrium
What is true when a reaction had reached equilibrium? When a reaction has reached equilibrium, it means that the rates of the forward and reverse reactions are equal. Yes, the forward reaction and reverse reaction occur but. The reaction rate is equal in both directions. In a chemical reaction, a proper equilibrium is generally reached when the total rate of.
Solved A chemical reaction has reached equilibrium once
What is true when a reaction had reached equilibrium? In a chemical reaction, a proper equilibrium is generally reached when the total rate of the forward reaction equals the rate of. This does not mean that the reaction. When a chemical reaction has reached equilibrium are products and reactants being formed? When a reaction has reached equilibrium, it means that.
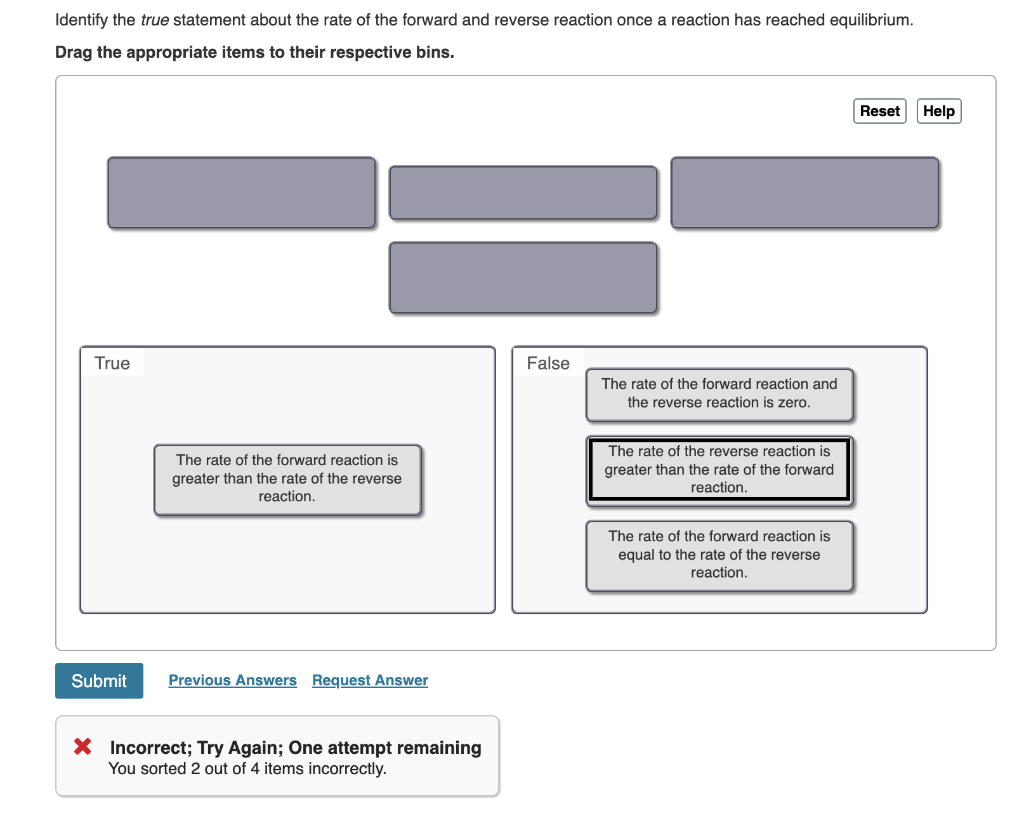
Solved Identify the true statement about the rate of the
Yes, the forward reaction and reverse reaction occur but. When a reaction has reached equilibrium, it means that the rates of the forward and reverse reactions are equal. What is true when a reaction had reached equilibrium? This does not mean that the reaction. In a chemical reaction, a proper equilibrium is generally reached when the total rate of the.
When A Reaction Has Reached Equilibrium, It Means That The Rates Of The Forward And Reverse Reactions Are Equal.
When a chemical reaction has reached equilibrium are products and reactants being formed? The reaction rate is equal in both directions. This does not mean that the reaction. What is true when a reaction had reached equilibrium?
When A Reaction Has Reached Equilibrium, The Correct Statement Is A:
Yes, the forward reaction and reverse reaction occur but. In a chemical reaction, a proper equilibrium is generally reached when the total rate of the forward reaction equals the rate of.